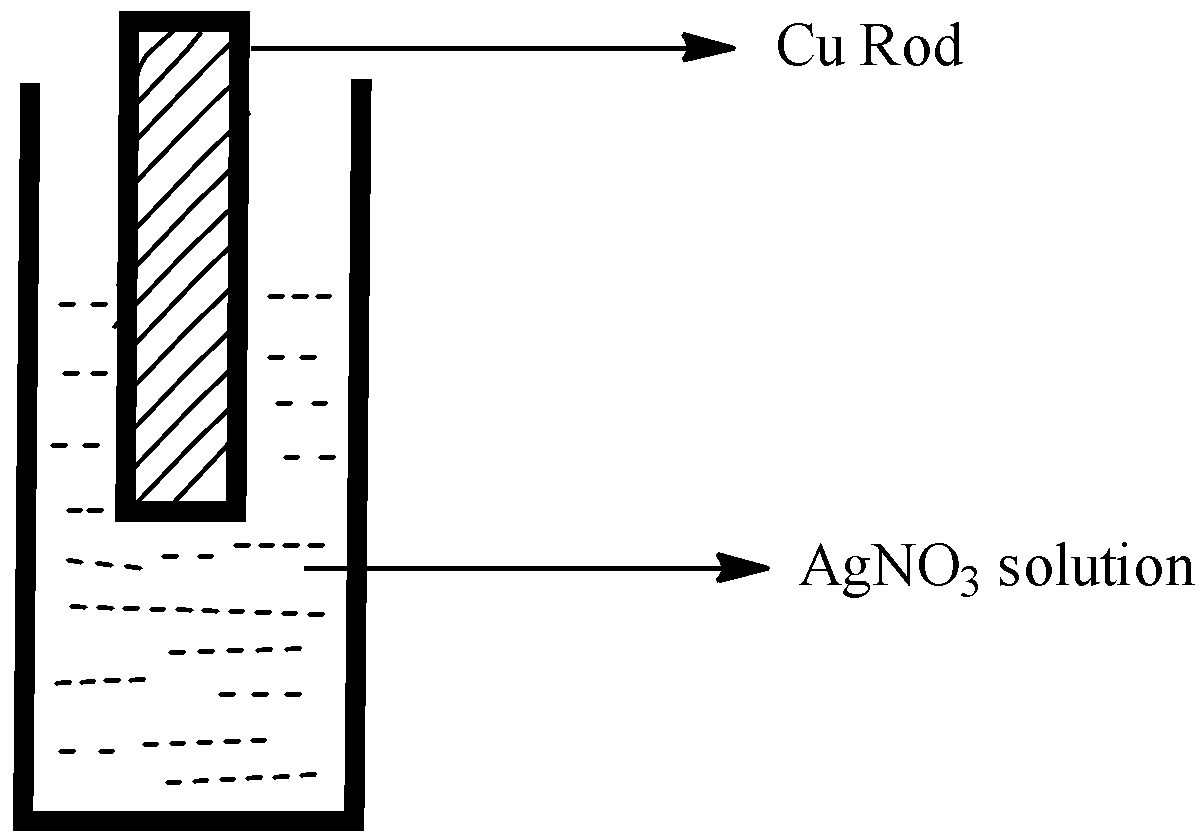
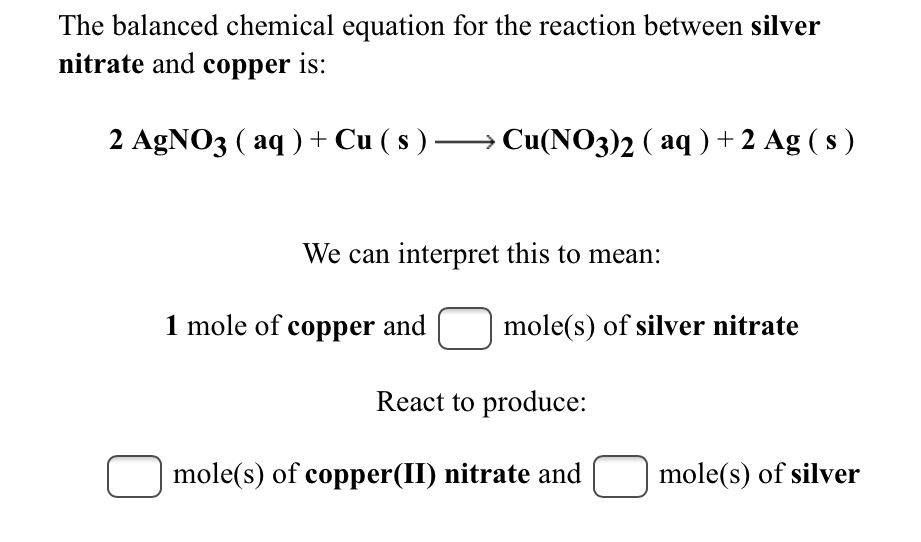
Observe the diagram showing a copper rod kept immersed in silver nitrate solution.a. What is the colour change of the solution?b. Write the balanced chemical equation for the reaction.
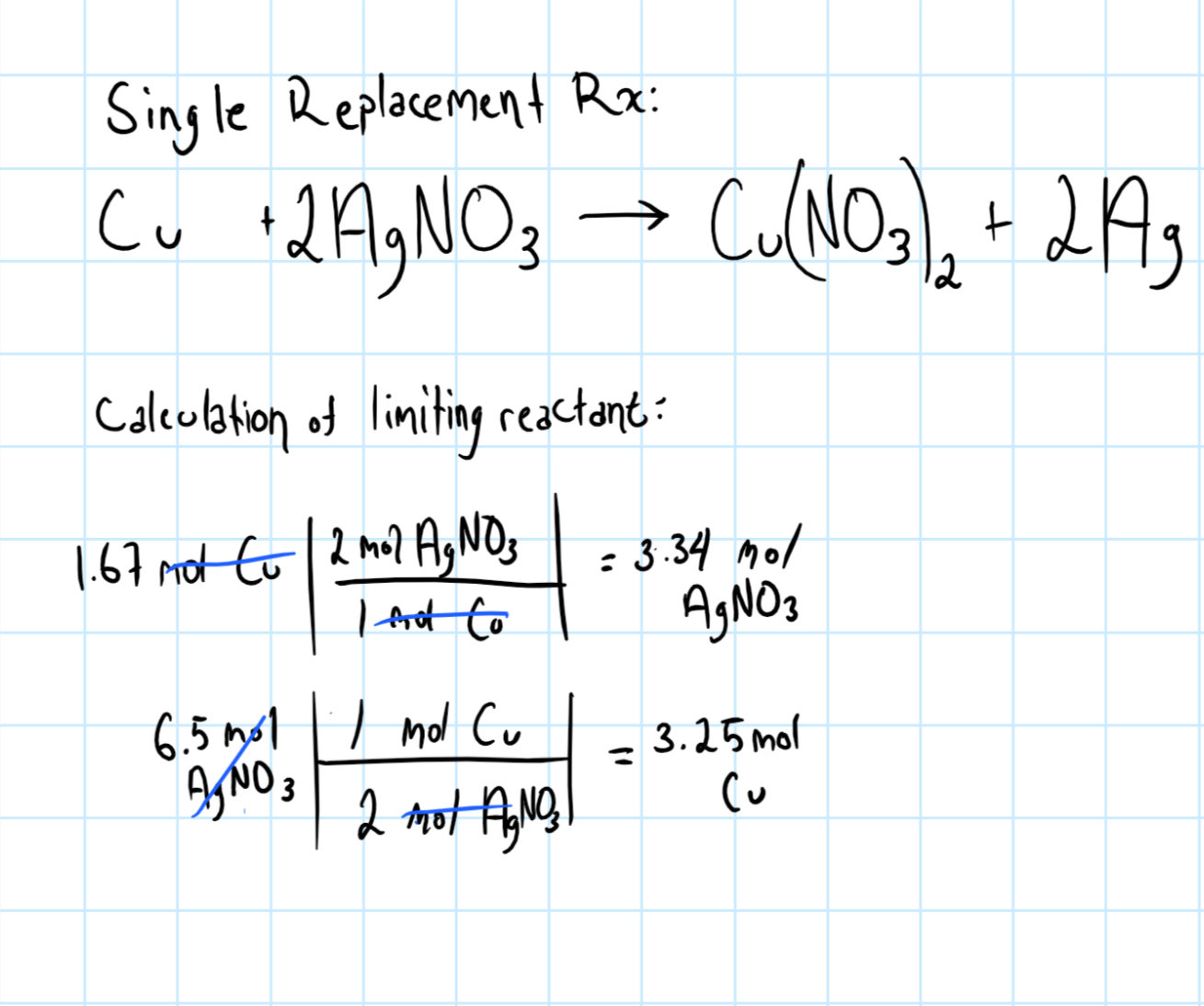
If 1.67 mol of copper and 6.5 mol of silver nitrate are available to react by single replacement, what is the limiting reactant? | Socratic

SOLVED: needed for this question. When silver nitrate reacts with chloride and copper(I) chloride, silver copper(D) nitrate are produced. The balanced equation for this reaction is: 2AgNO; (aq) + CuClz (s) 2AgCl(s) +
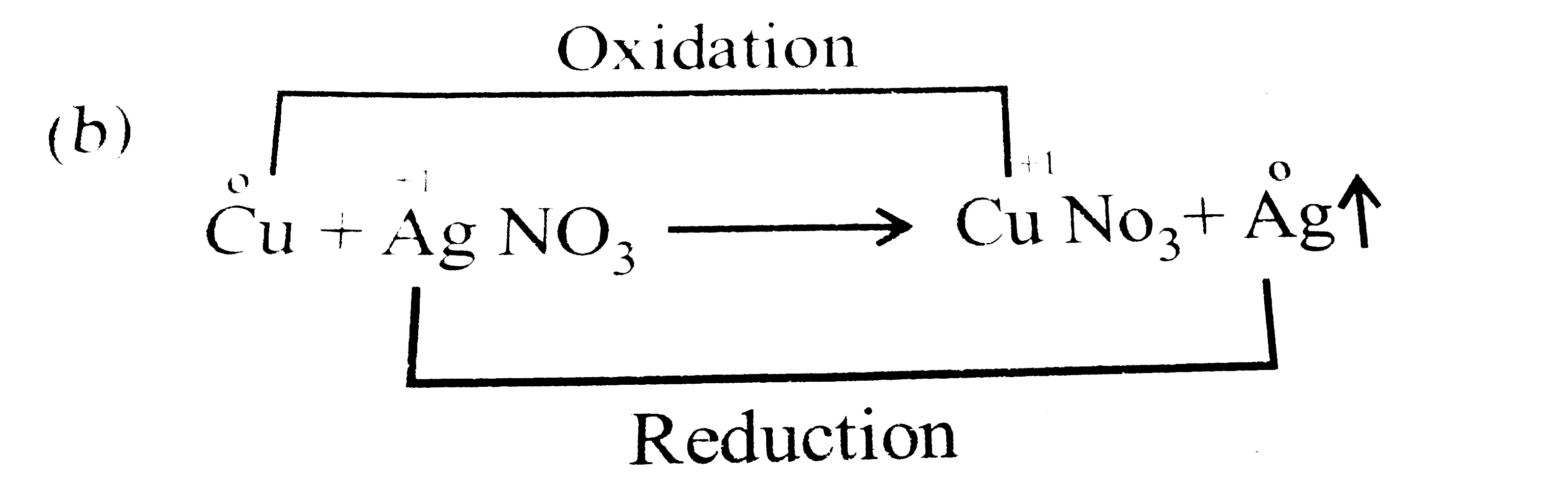
When a copper rod is dipped in aqueous silver nitrate solution , the colour of the solution changes to blue . This is because

a) The recovery of silver nitrate solution involves displacement by copper metal. Write the - YouTube

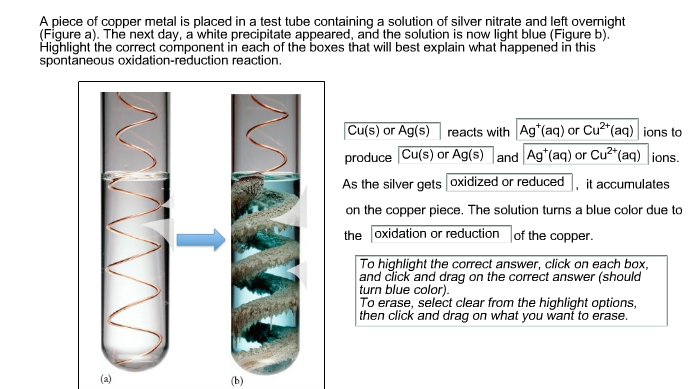
A student performs an experiment in which he dipped a copper coil to the silver nitrate solution.Which of the following is the correct observation related to this experiment?