
Utilising a Virtual Central Laboratory Approach to Balance the Delivery, Budget and Risk for Clinical Trials - Simbec Orion

Fifth Annual Trial Advocacy Summer Institute Held at Central Courthouse – Seventeenth Judicial Circuit of Florida
Study flow diagram. CENTRAL, Cochrane Central Register of Controlled... | Download Scientific Diagram

The Medical Device Regulation: Why a Delay in Implementation, and What is The Impact on Your Clinical Trials? - GCP Central
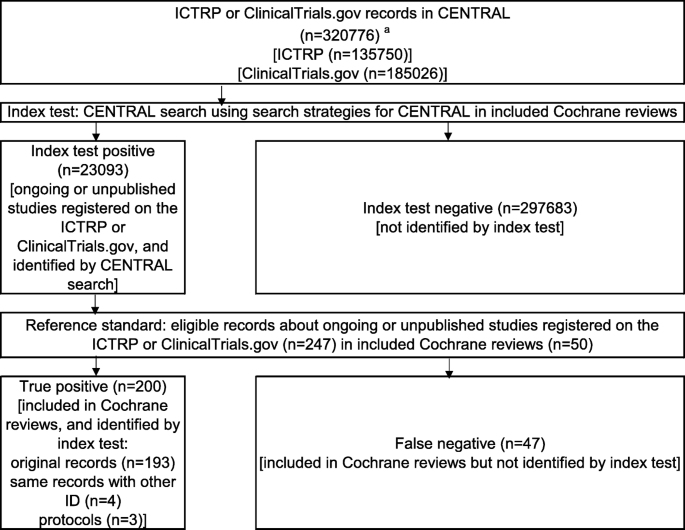
Using the Cochrane Central Register of Controlled Trials to identify clinical trial registration is insufficient: a cross-sectional study | BMC Medical Research Methodology | Full Text
Central Lab Role Clinical Trials Quality Data Capture | PowerPoint Slide Templates Download | PPT Background Template | Presentation Slides Images

Monitoring Site vs Central Discordance in Oncology Clinical Trials: The Value of a Unified Imaging/EDC Platform | Medidata Solutions
RESOLVED Advisory: Customers are unable to create trials on Sophos Central Web (different than Sophos Central Partner Dashboard)
















