The crystal structure of anhydrous nitrates and their complexes. II. The 1:1 copper(II) nitrate–nitromethane complex - Duffin - 1966 - Acta Crystallographica - Wiley Online Library

Many nitrate salts can be decomposed by heating. For example, blue, anhydrous copper(II) nitrate produces the gases nitrogen dioxide and oxygen when heated. In the laboratory, you find that a sample of

Innovating Science Anhydrous Copper (II) Sulfate, 500g - Reagent Grade – Excellent for Biochemistry & Organic Chemistry Experiments - The Curated ...
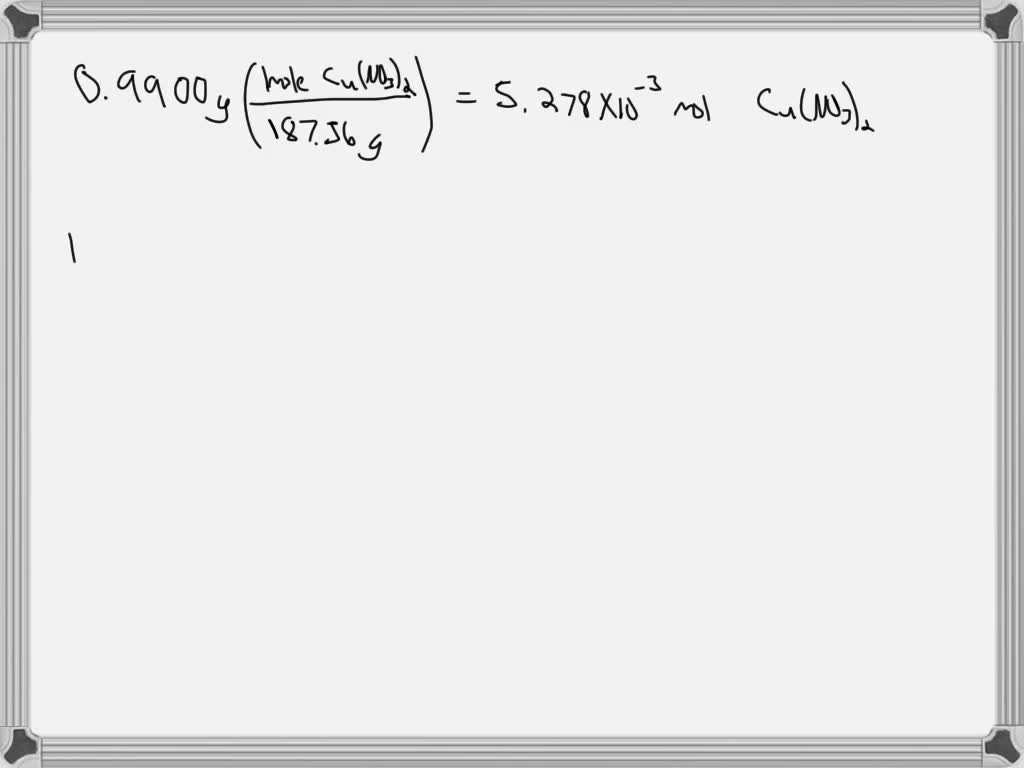
SOLVED: A sample of copper (II) nitrate hydrate has a mass of 1.278 g. The sample is heated until all water is removed resulting in a final mass of anhydrous copper (II)