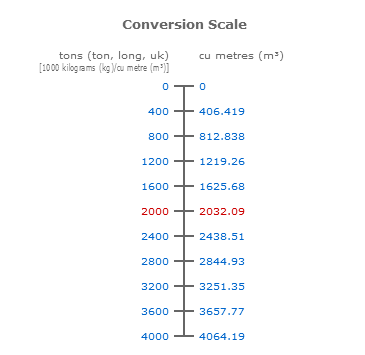
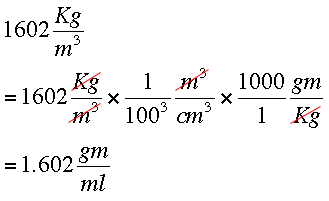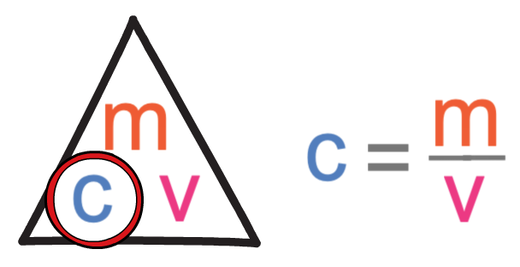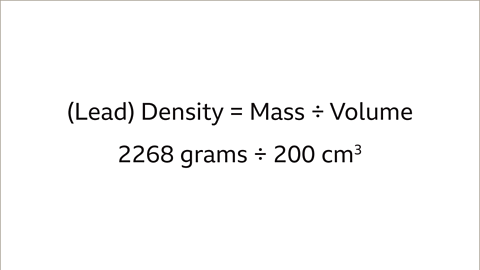SOLVED: Use molar volume to calculate each of the following at STP: a. the number of grams of Ne contained in 11.2 L of Ne gas b. the volume, in liters, occupied
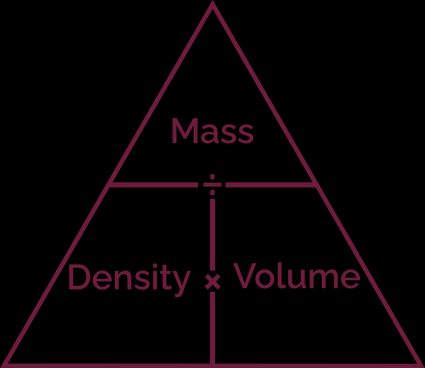
Solved: Gathered Data: Density = 1.80 g/mL Volume=0.32 L Required: mass in grams Solution: Mass=(D [Chemistry]

An empty beaker weighs 42.75 grams. When completely filled with water, the beaker and its contents have a total mass of 399.95 grams. What volume does the beaker hold? Use d =