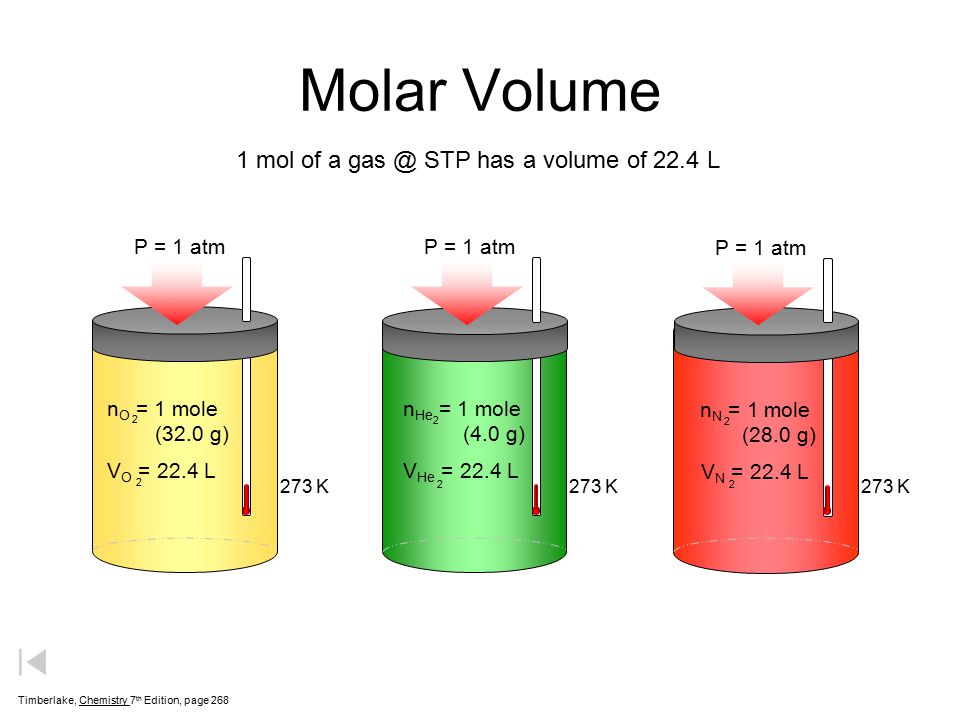
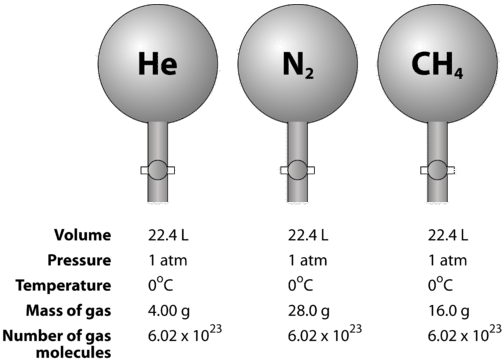
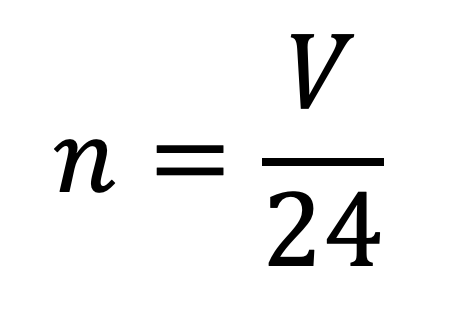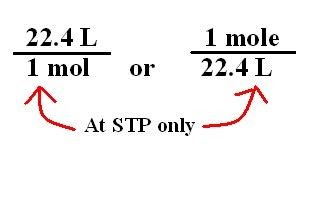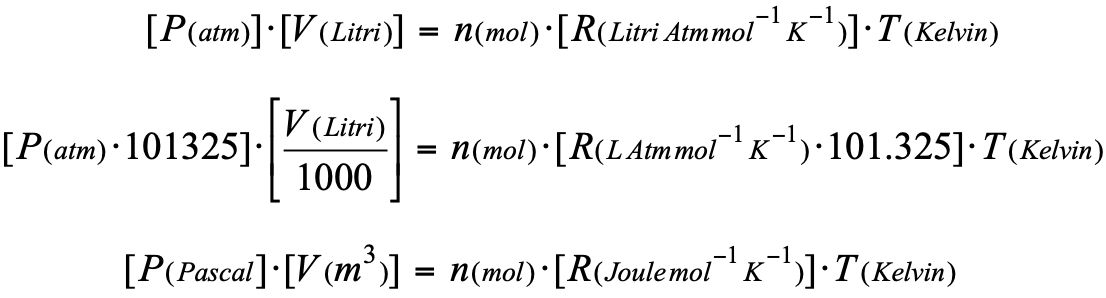Volume molare di un gas - Il significato del numero 22,4 litri - Teoria + Esercizi svolti - Il Prof Che Ci Piace

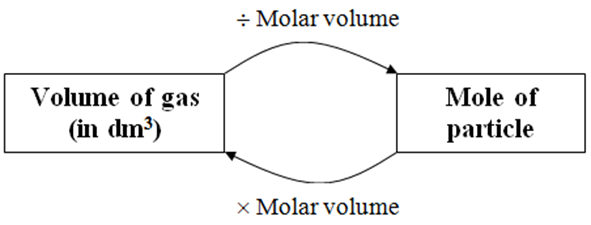
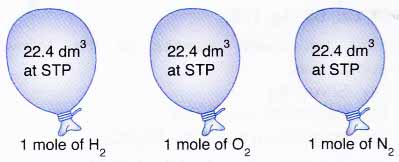
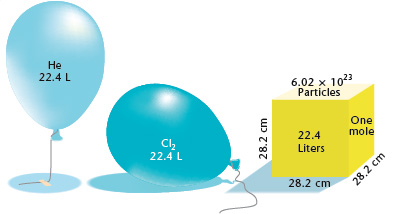
Molar volume is the volume occupied by 1 mol cif any (ideal) gas at standard temperature and pressure (STP. 1 atm. pressure 0°C). Show that it is 22.4 litres - CBSE Class

SOLVED: 4. What volume is occupied by 1 mol of an ideal gas at STP (standard temperature and pressure means t=0°C and p=1atm)? Ans.: V=22.418 × 10^-3 m^3 = 22.418 L 5.
Determine the volume of 1 mole of any gas at s. T. P., assuming it behaves like an ideal gas? - Sarthaks eConnect | Largest Online Education Community