
Table 1 from The MDS-UPDRS Part II (motor experiences of daily living) resulted useful for assessment of disability in Parkinson's disease. | Semantic Scholar
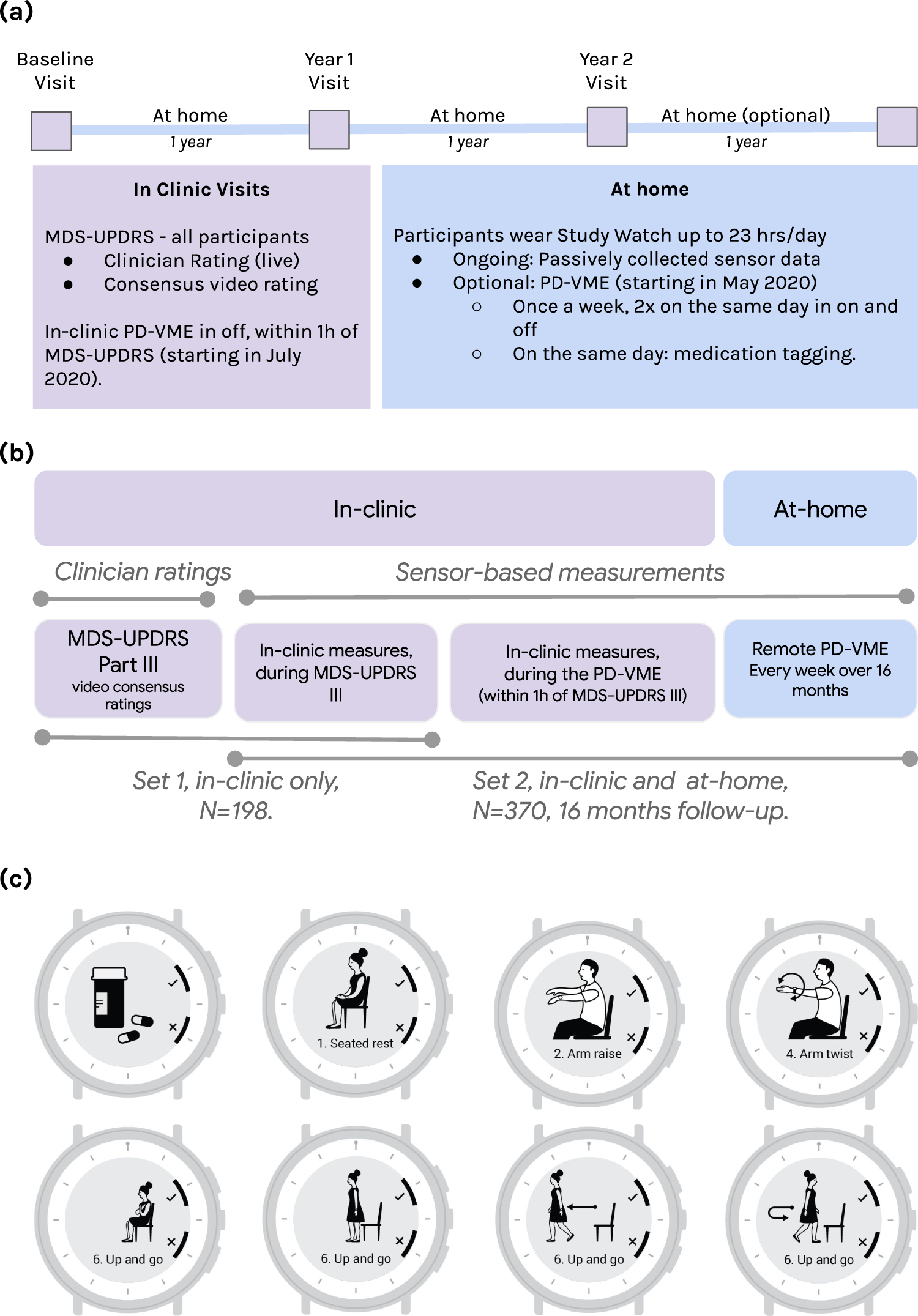
Virtual exam for Parkinson's disease enables frequent and reliable remote measurements of motor function | npj Digital Medicine

Parkinson's Disease Center at BIDMC on X: "A GLP1 agonist, NLY01, did not meet the primary outcome in a ph2 study in #PD, but a pre-specified analysis showed significant benefits for both
Correlation Between PDQ-39 and MDS-UPDRS Scores in a Phase 3 Randomized Controlled Trial of Patients with Parkinson's Disease - MDS Abstracts

Some exciting GLP-1 drug new randomized trial evidence for potential disease-modifying impact on #Parkinson's disease (there are none). No progression of motor disability in the Rx group for 12 months. https://nejm.org/doi/full/10.1056/NEJMoa2312323
![PDF] SPES/SCOPA and MDS-UPDRS: formulas for converting scores of two motor scales in Parkinson's disease. | Semantic Scholar PDF] SPES/SCOPA and MDS-UPDRS: formulas for converting scores of two motor scales in Parkinson's disease. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3b30592ca82ccdf0a1c52a5e8183eeb27adcb6ba/2-Table1-1.png)
PDF] SPES/SCOPA and MDS-UPDRS: formulas for converting scores of two motor scales in Parkinson's disease. | Semantic Scholar

Frontiers | Clinically important change on the Unified Dyskinesia Rating Scale among patients with Parkinson's disease experiencing dyskinesia

Figure 1 from A MDS-UPDRS Part II (motor experiences of daily living) score to predict recurrent falls in people with Parkinson's disease | Semantic Scholar
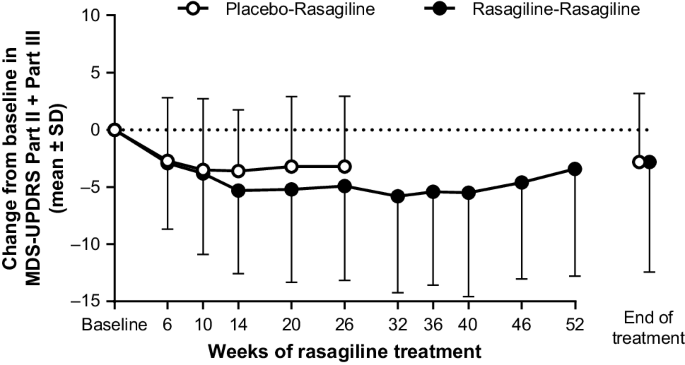
Long-term, open-label, phase 3 study of rasagiline in Japanese patients with early Parkinson's disease | Journal of Neural Transmission
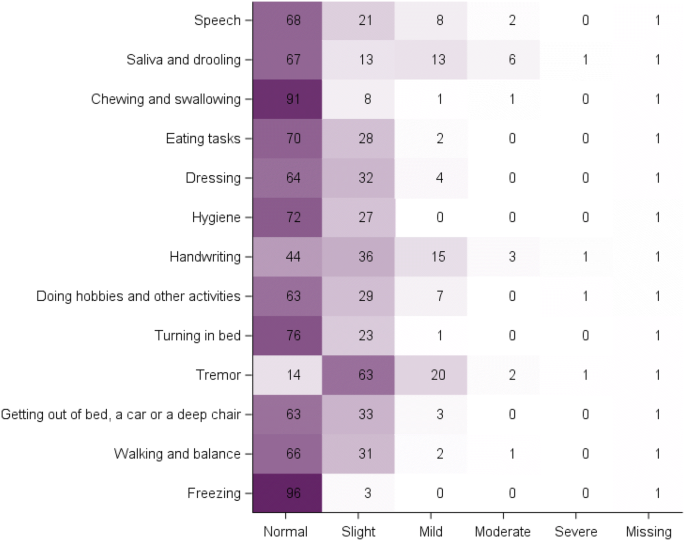
Does the MDS-UPDRS provide the precision to assess progression in early Parkinson's disease? Learnings from the Parkinson's progression marker initiative cohort | Journal of Neurology

A Phase II study to evaluate the safety and efficacy of prasinezumab in early Parkinson's disease (PASADENA): rationale, design and baseline data | medRxiv
Weighting MDS-UPDRS Part II items for optimal sensitivity to Parkinson's disease progression using Parkinson's Progression M


![UPDRS Part I questions 1 and 2 [2]. | Download Scientific Diagram UPDRS Part I questions 1 and 2 [2]. | Download Scientific Diagram](https://www.researchgate.net/publication/351975927/figure/fig1/AS:1080189173923849@1634548576236/UPDRS-Part-I-questions-1-and-2-2.jpg)








