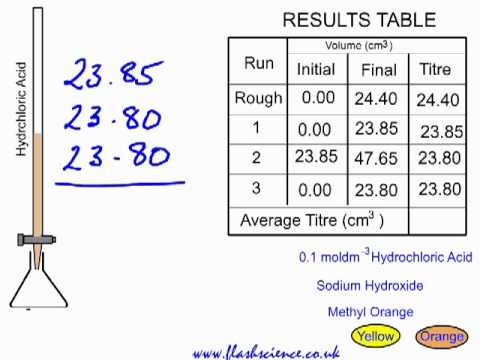
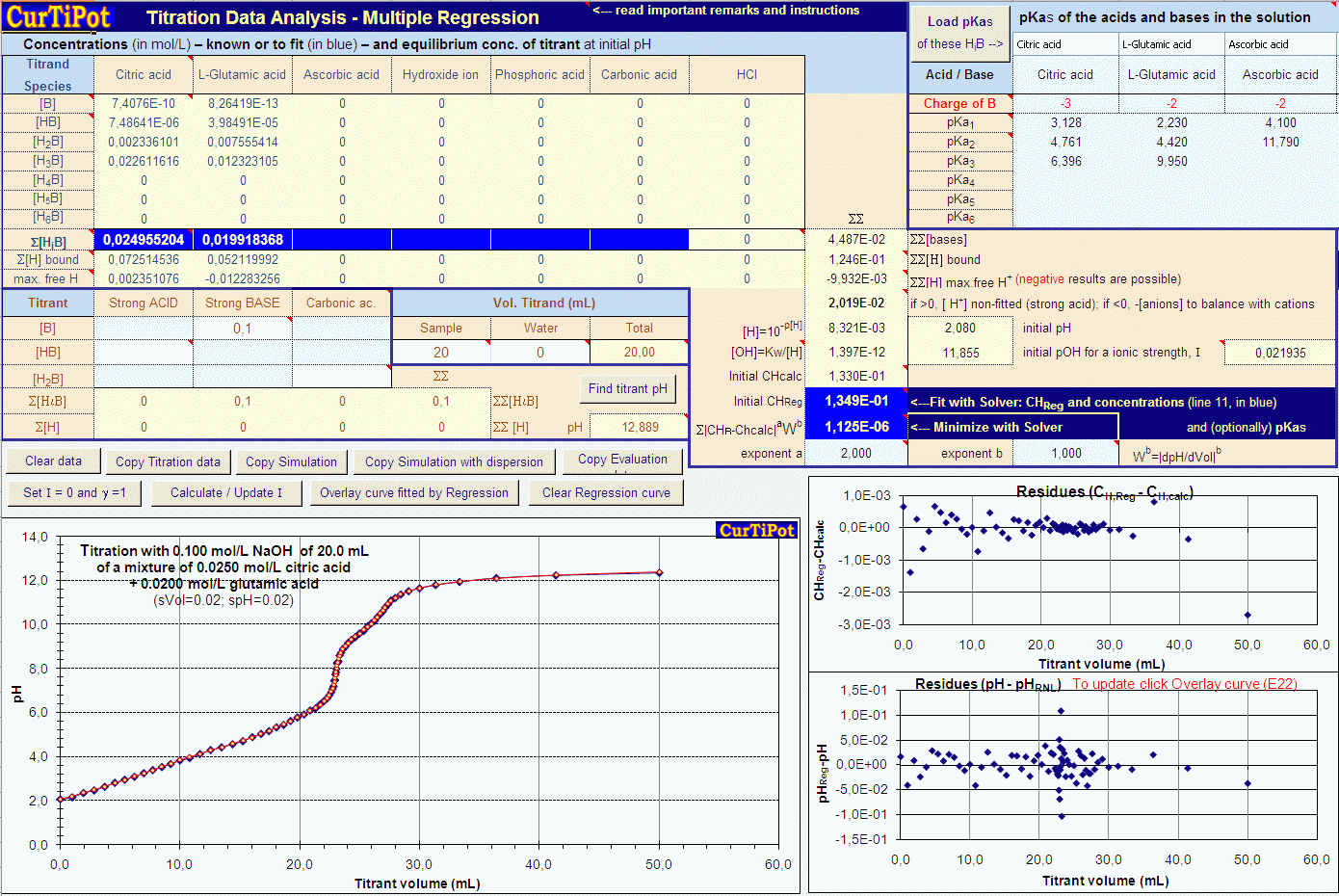
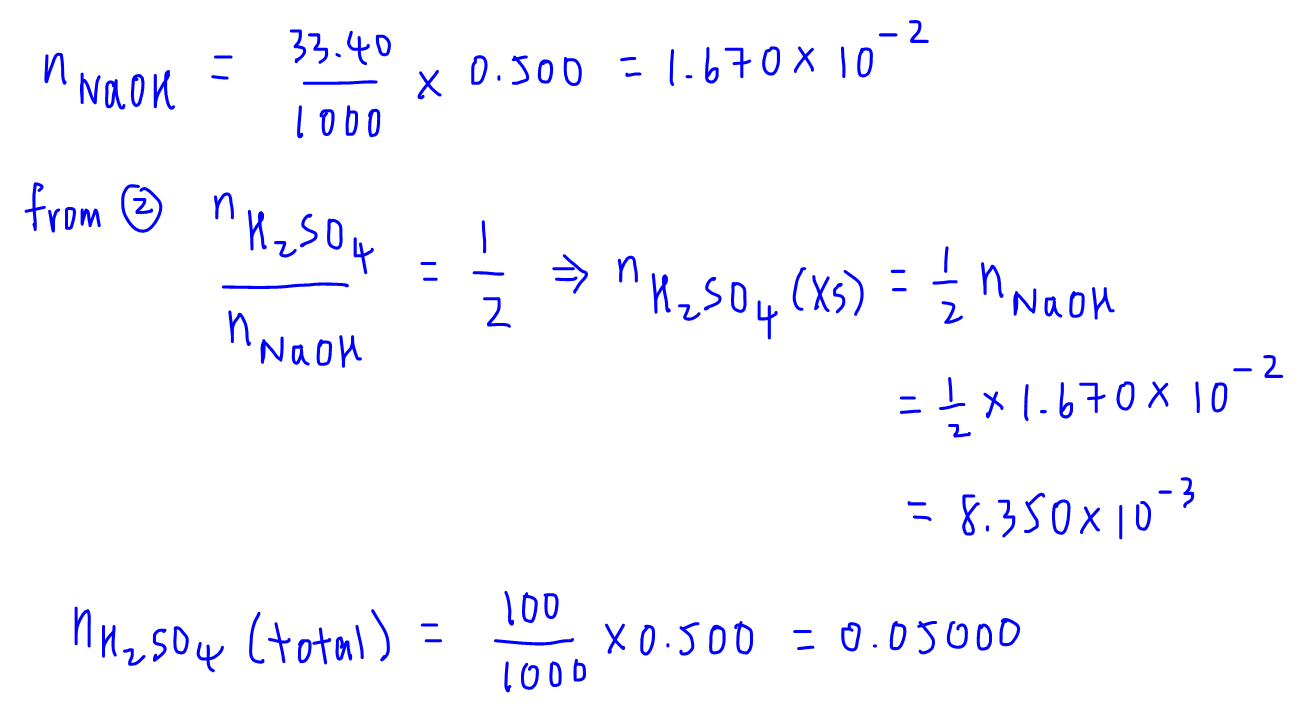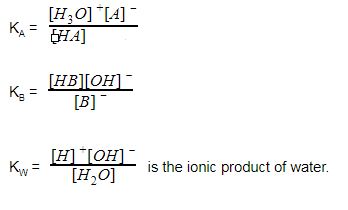Acid-alkali volumetric titrations calculating concentrations from experimental results, apparatus, indicator, conical flask, pipette, burette gcse chemistry Calculations antacid indigestion tablet igcse KS4 science A level GCE AS A2 O Level practice ...
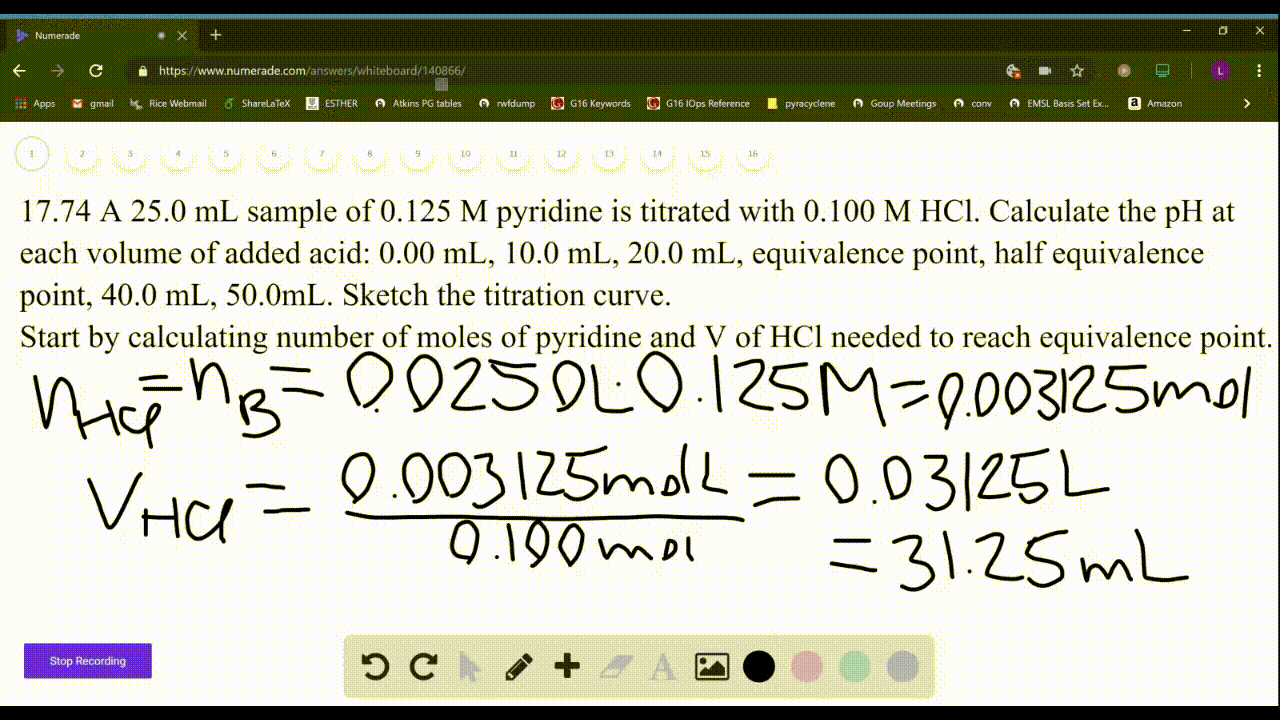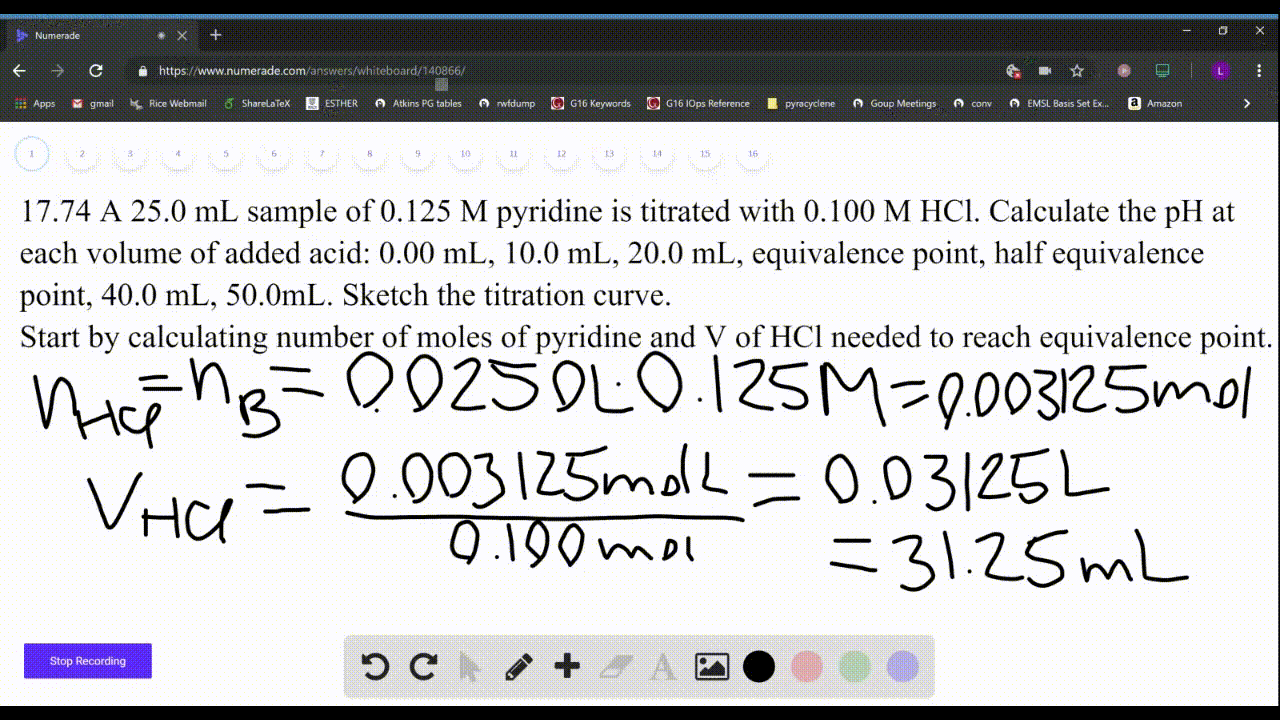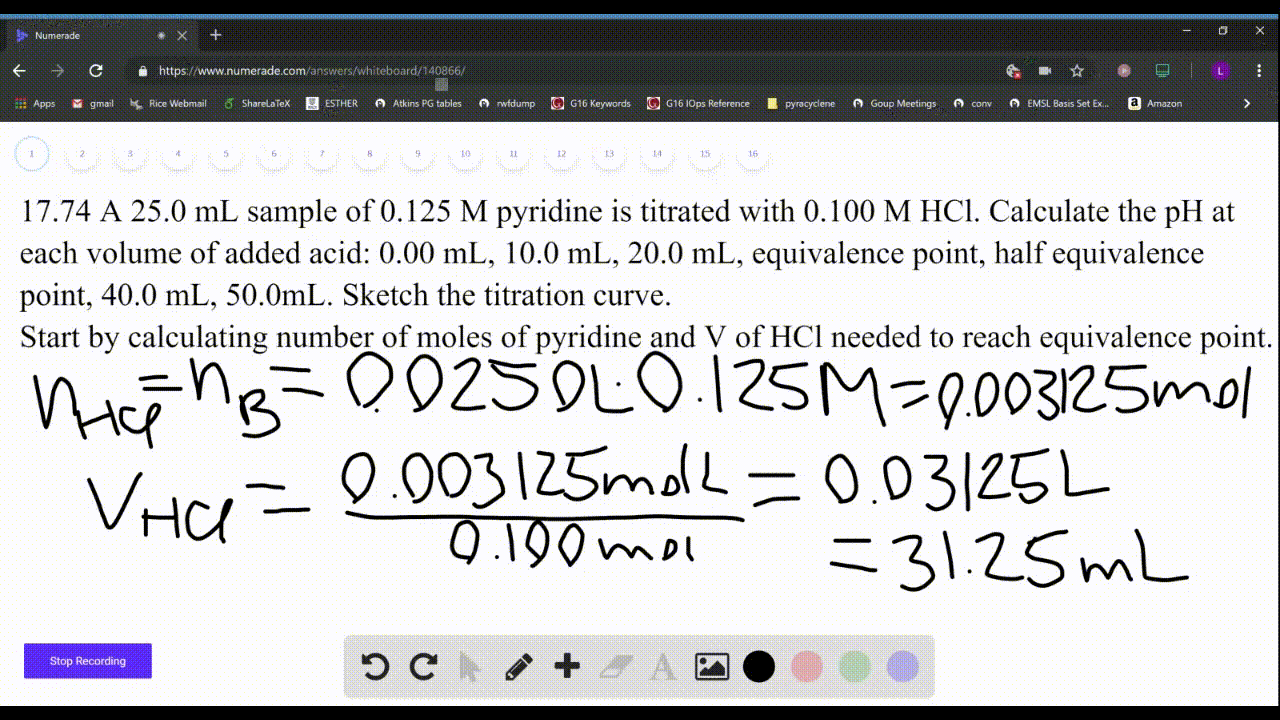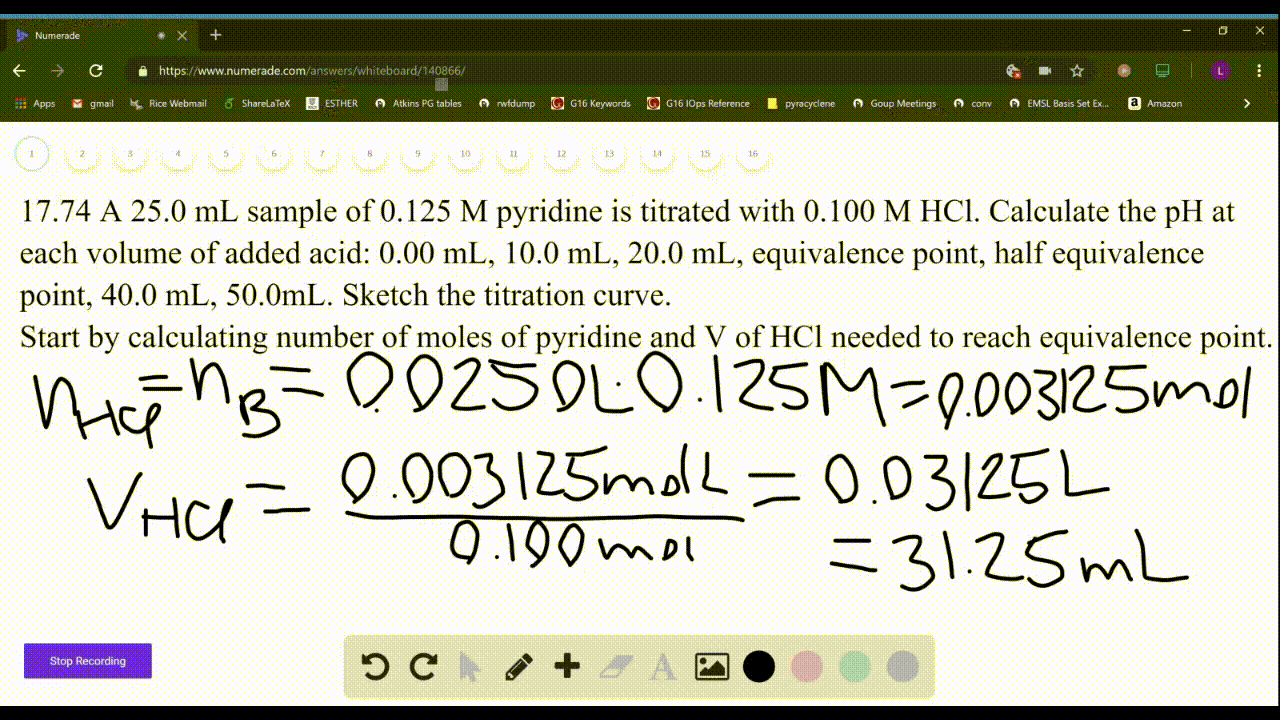
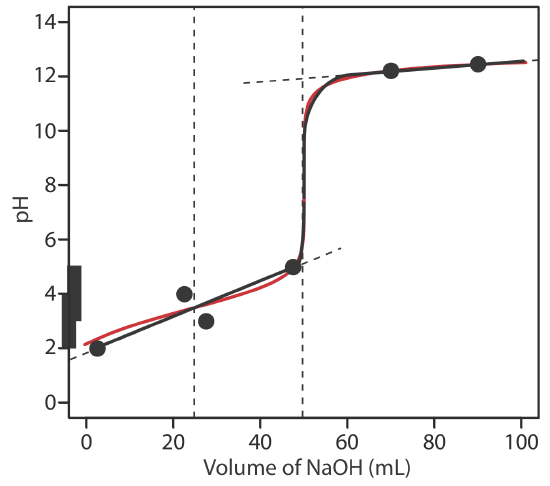
SOLVED: A 25.0 -mL sample of 0.125 M pyridine is titrated with 0.100 M HCl. Calculate the pH at each volume of added acid: 0 mL, 10 mL, 20 mL , equivalence