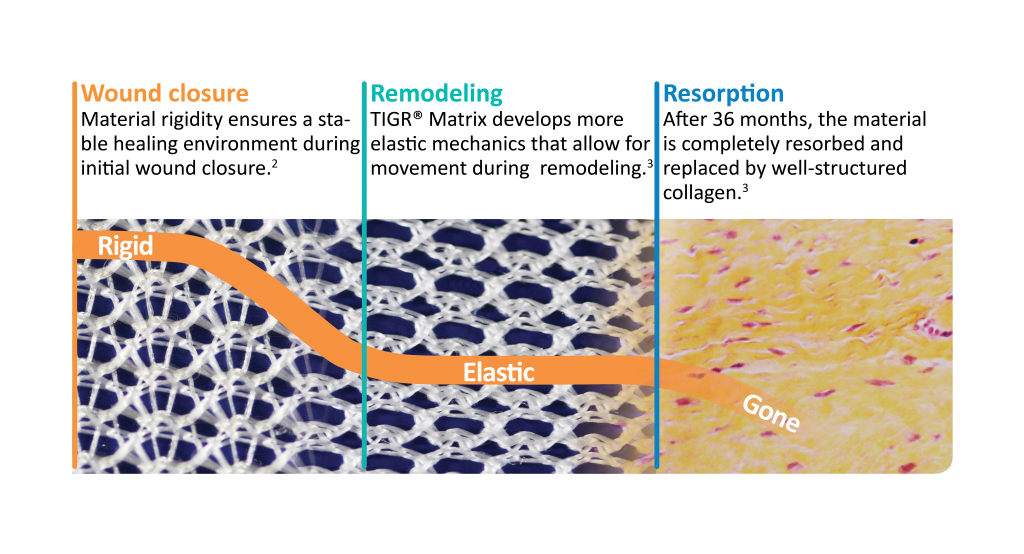
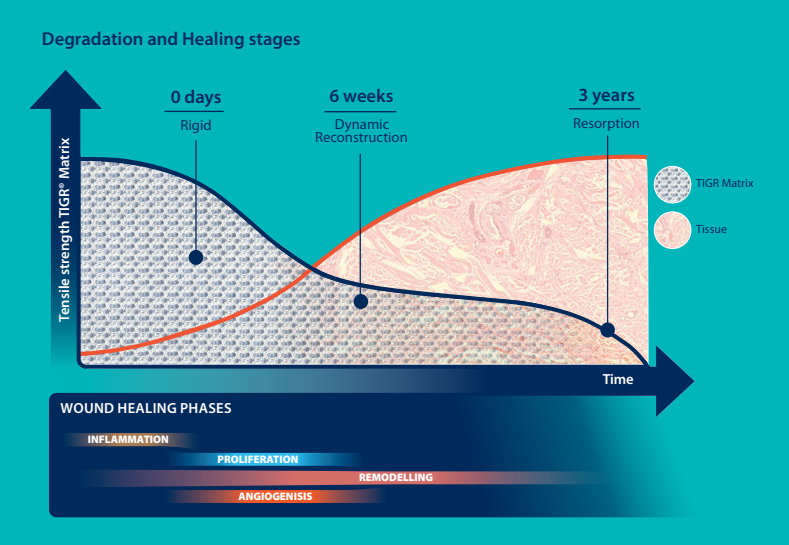
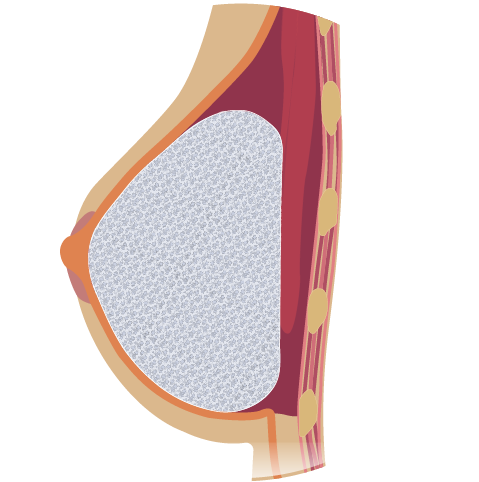
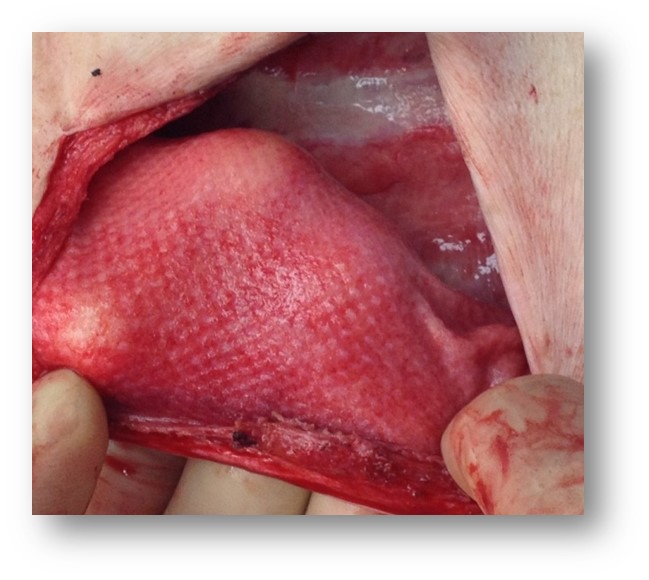
Novus Scientific receives CE Mark Approval for TIGR® Matrix Surgical Mesh - the World's First Long-Term Resorbable Synthetic Mesh. | TIGR® Matrix

Full article: TIGR® matrix surgical mesh – a two-year follow-up study and complication analysis in 65 immediate breast reconstructions

12 Month TRAM Data Collated Using TIGR® Matrix Surgical Mesh in a Randomized Controlled Study Presented at MAPACS 2012. | TIGR® Matrix

Novus Scientific Announces the Worlds 1st Long-Term Absorbable Mesh as an Alternative to Non-Absorbable Synthetic Mesh. | TIGR® Matrix

Novus Scientific presents initial results of the First-in-Man study for TIGR® Matrix Surgical Mesh – The world's 1st long-term resorbable synthetic mesh | Novus Scientific