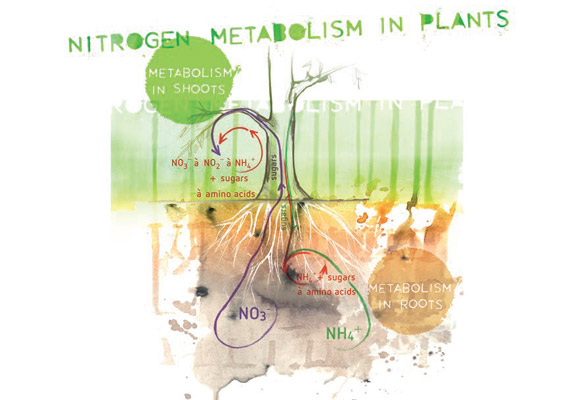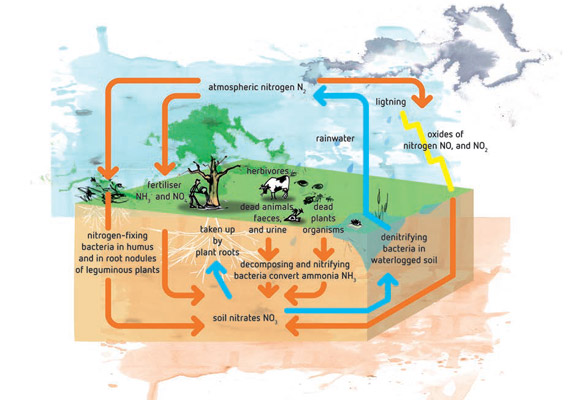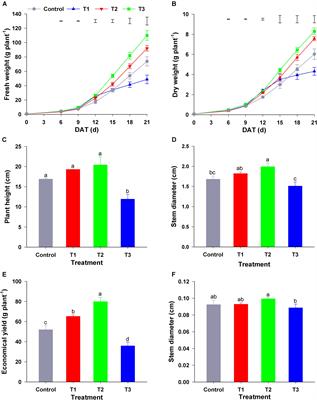
Frontiers | Appropriate NH4+/NO3– Ratio Triggers Plant Growth and Nutrient Uptake of Flowering Chinese Cabbage by Optimizing the pH Value of Nutrient Solution

SOLVED: Ammonium nitrate (NH4NO3) is a salt used in fertilizers and in making matches. The salt is made by the reaction of ammonia (NH3) and nitric acid (HNO3). Calculate the pH of

SOLVED: Ammonium nitrate (NHANO3) is a salt used in fertilizers and in making matches. Calculate the pH of a 0.15 M solution of ammonium nitrate: (Kb for NHz is 1.8 x 10-5)

Ammonium nitrate, NH4NO3, is a salt formed from the neutralization of the weak base ammonia with the strong acid nitric acid. Given that the value of Kb for ammonia is 1.8 x
Particle phase fraction of total nitrate, ε(NO − 3 ) (blue curve), and... | Download Scientific Diagram

Effects of pH and H2O2 on ammonia, nitrite, and nitrate transformations during UV254nm irradiation: Implications to nitrogen removal and analysis - ScienceDirect
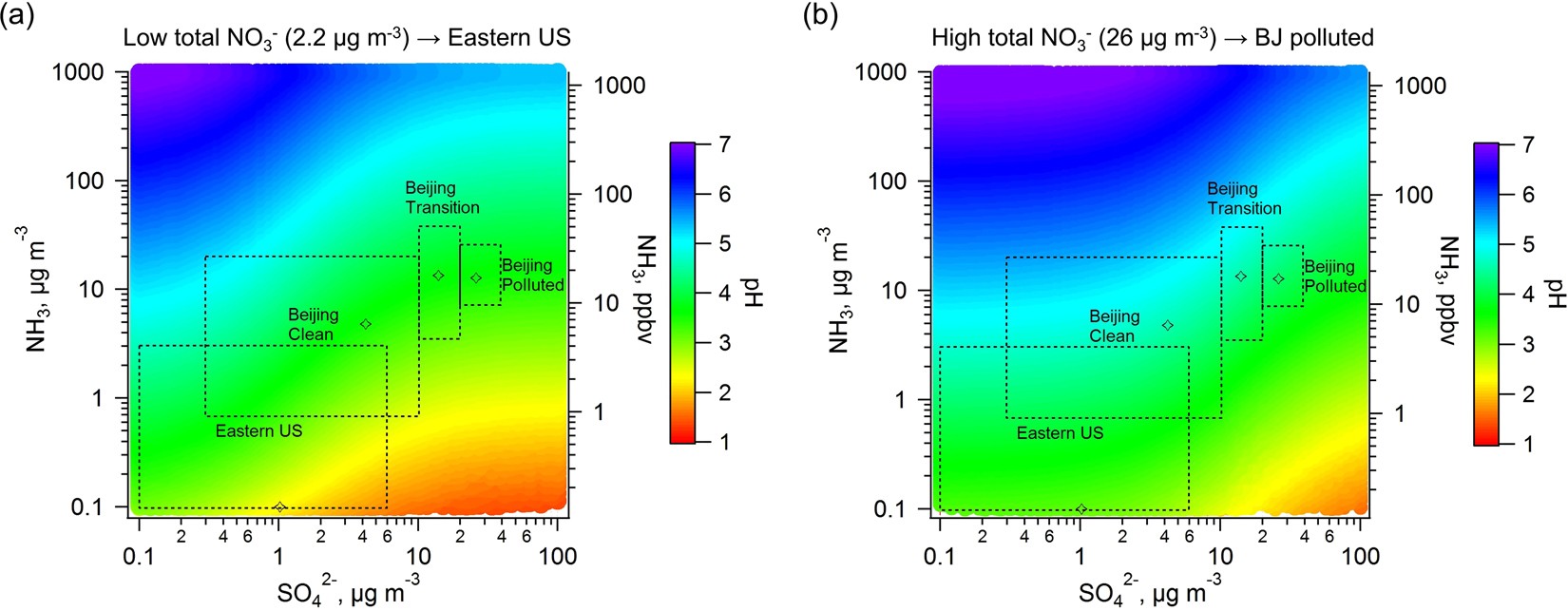
High levels of ammonia do not raise fine particle pH sufficiently to yield nitrogen oxide-dominated sulfate production | Scientific Reports