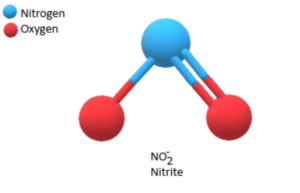
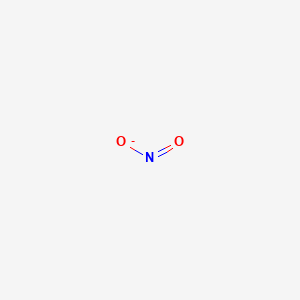
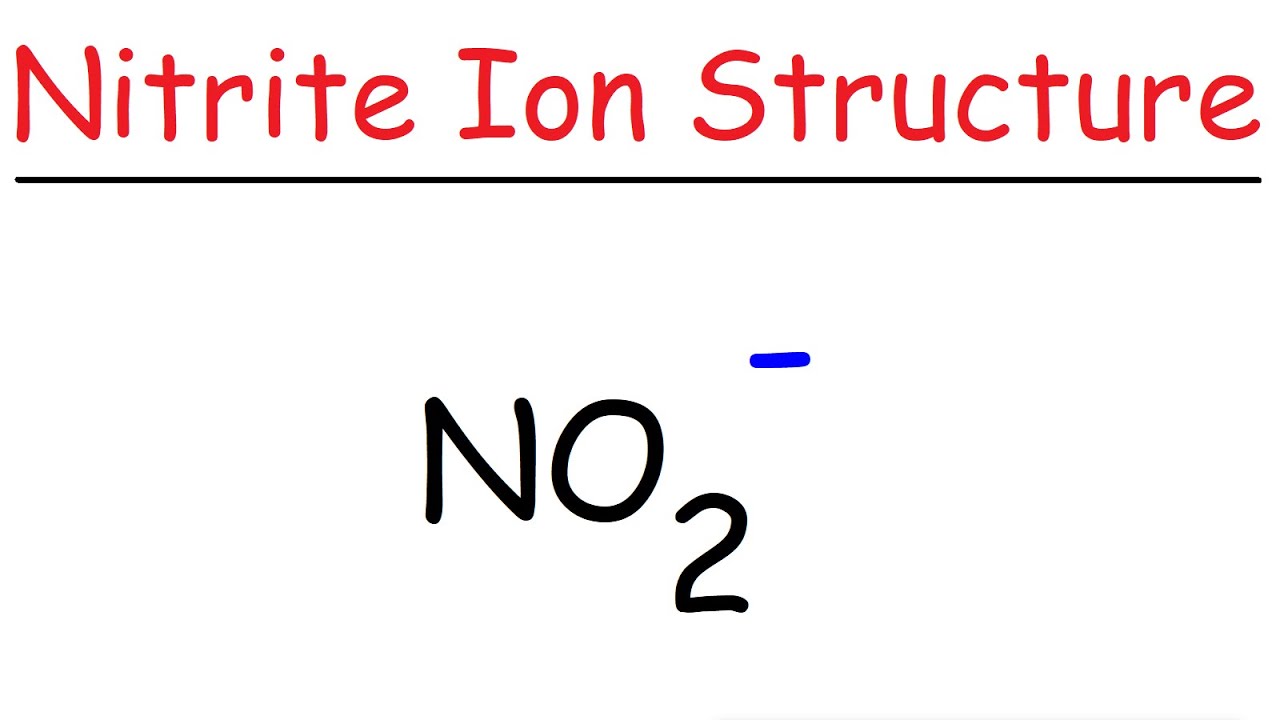
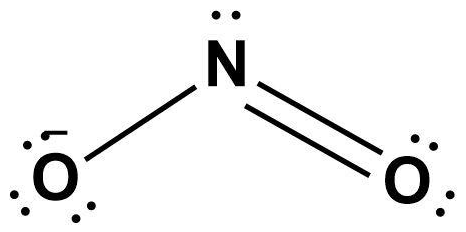
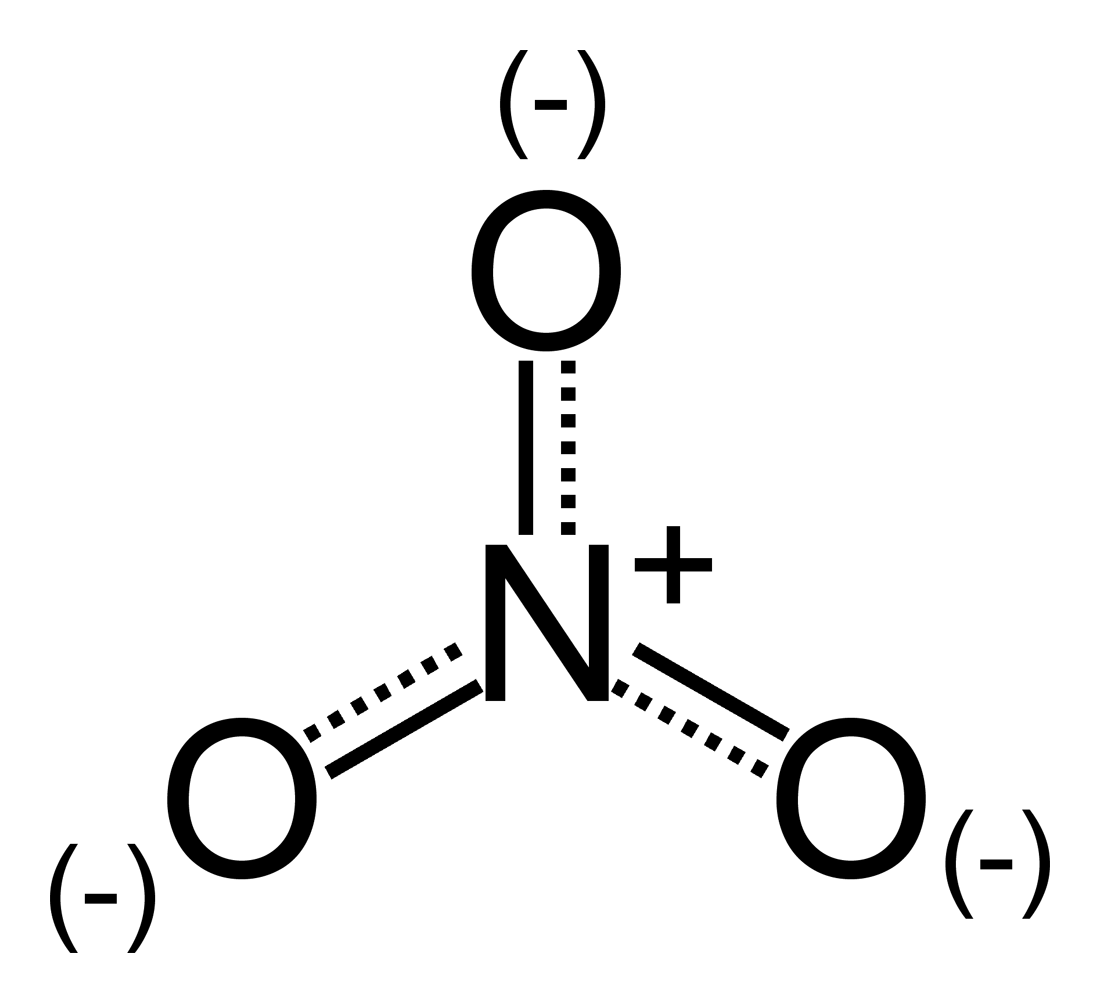Use Lewis theory to draw the structure of the nitrite polyatomic ion. What would you find surrounding the central atom of this ion? A. two bonding and one unshared pair of electrons.

For the nitrite ion (NO2-), identify the following. a. How many total lone pairs are in the ion? b. What is the molecular shape of the ion? c. How many resonance structures

A diagram of the proposed major pathways for nitrate ion transport and... | Download Scientific Diagram

The number of valence electrons in the nitrite ion is: A) 22 B) 16 C) 23 D) 18 E) 24 | Homework.Study.com

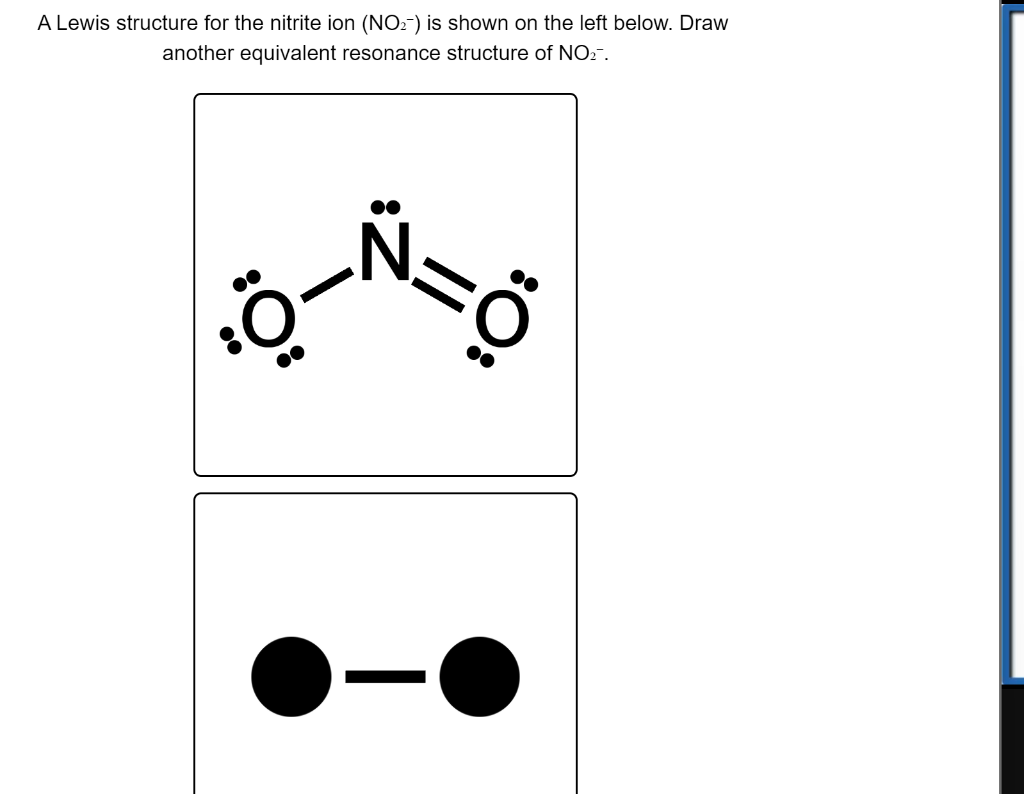
a. Draw Lewis structure for the nitrite ion, NO2-. b. Draw Lewis DOT structure for the nitrite ion, NO2-. Use square brackets to denote the overall charge. | Homework.Study.com
![Nitrite Ion Standard Solution (NO2- 1000)・140-06451・146-06453[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Nitrite Ion Standard Solution (NO2- 1000)・140-06451・146-06453[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/05/146-06453.png)