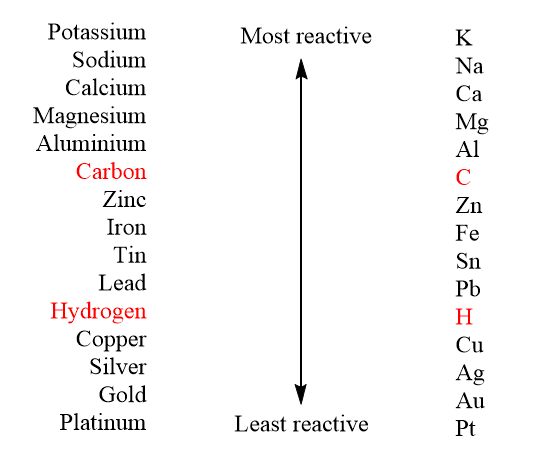
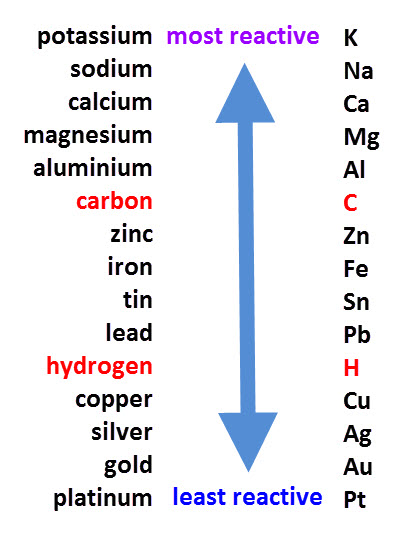
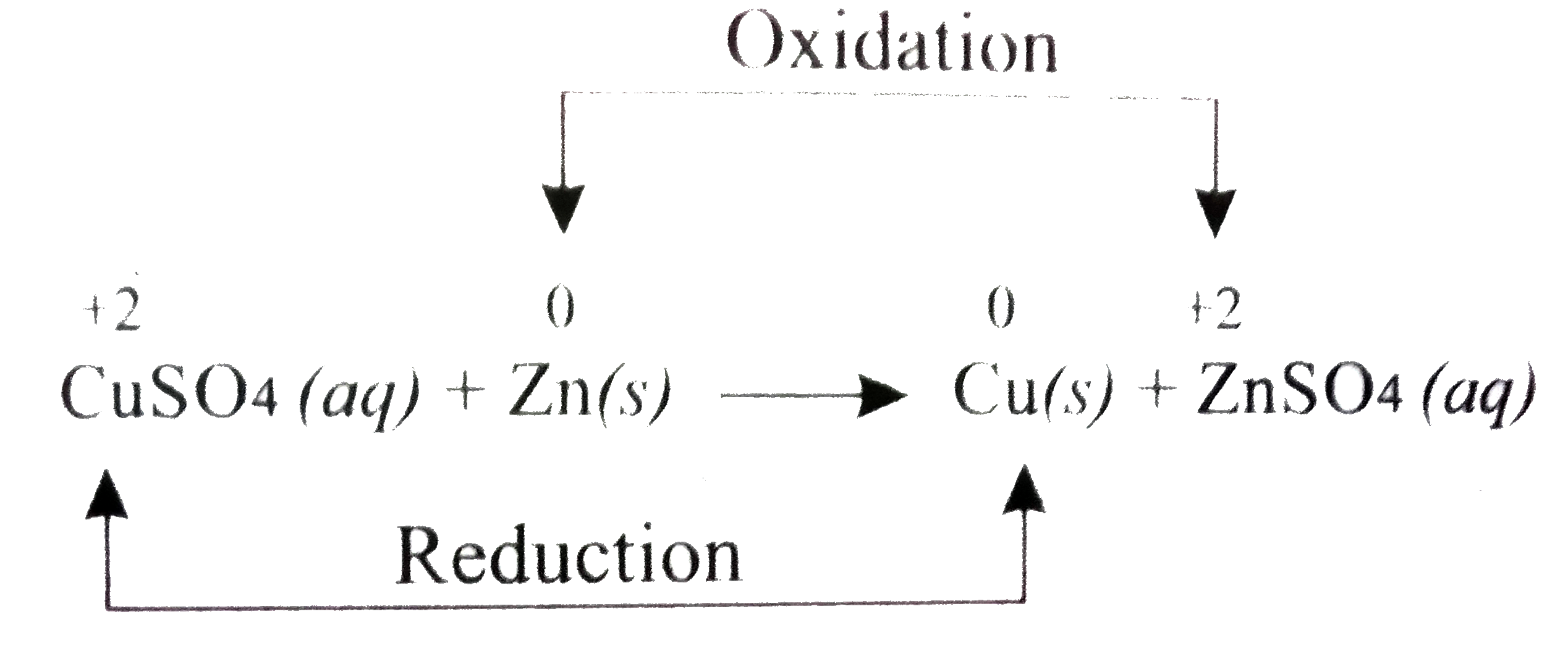
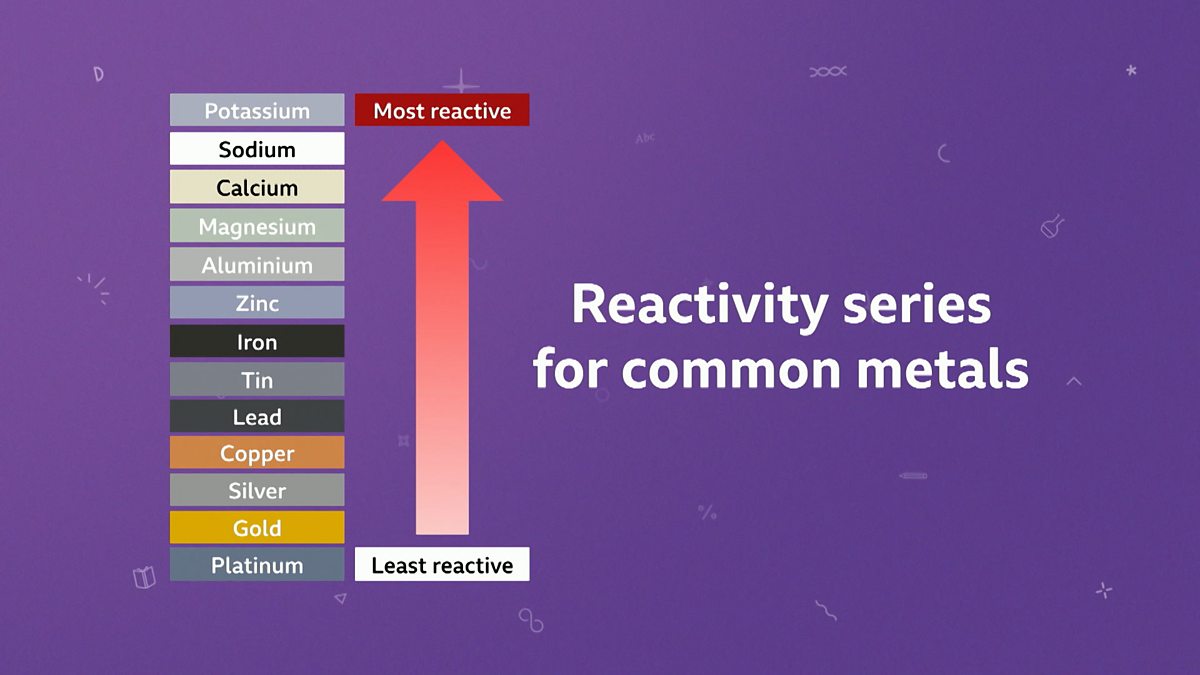
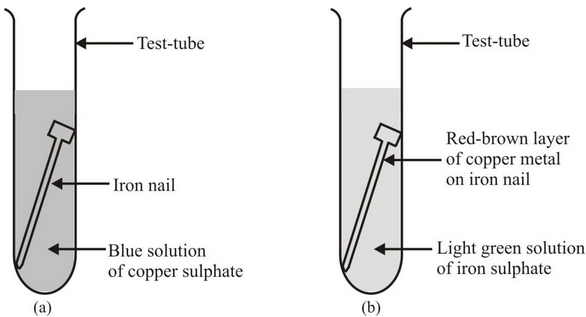
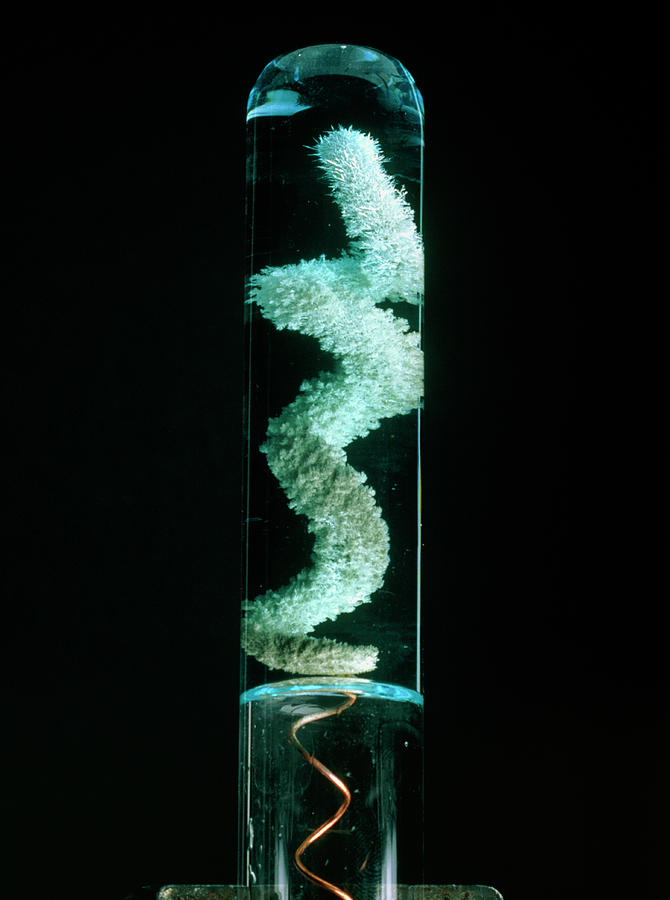
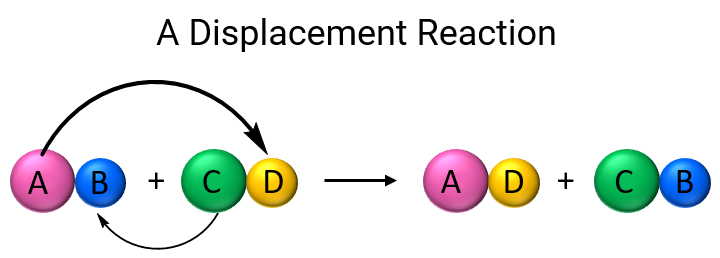
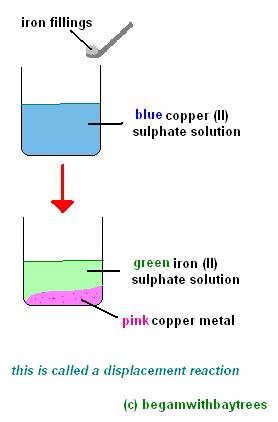
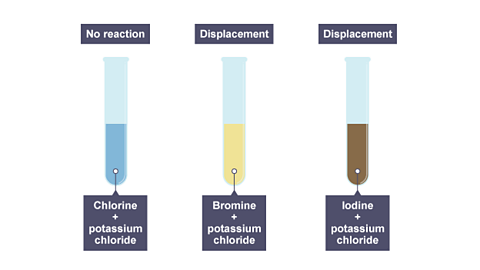
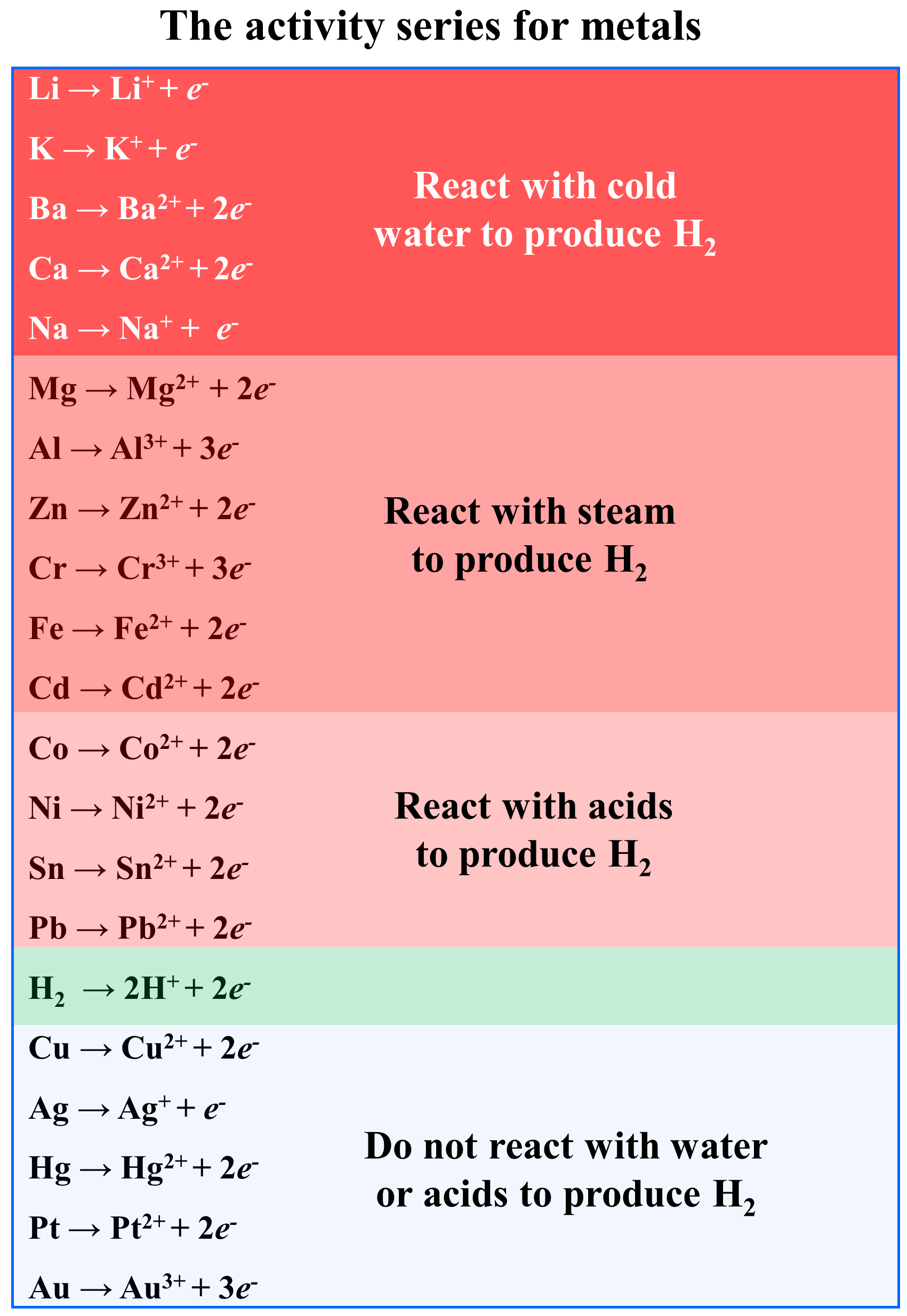please explain the displacement reaction of metals and non-metals - Chemistry - Redox Reactions - 4575986 | Meritnation.com
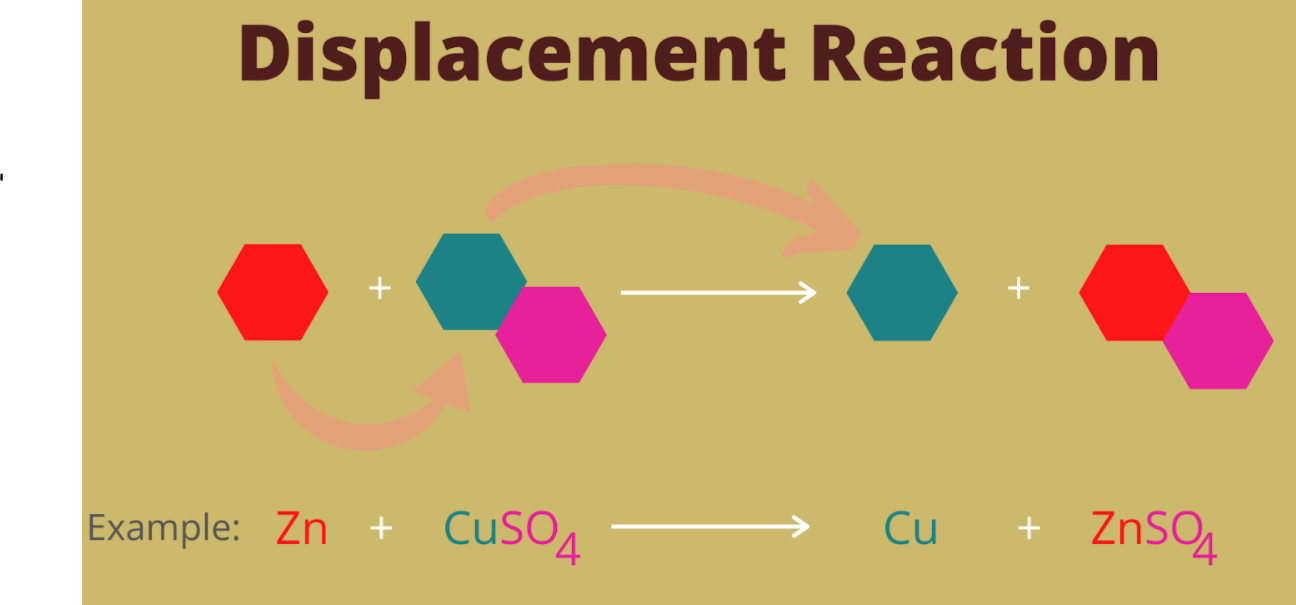
Which displacement reaction will not occur among the following reactions?A) ${ Zn+ } { CuSO }_{ 4 }$B) ${ Fe+ } { CuSO }_{ 4 }$C) ${ Cu+ } { FeSO }_{ 4 }$D) ${ Mg+ } { CuSO }_{ 4 }$