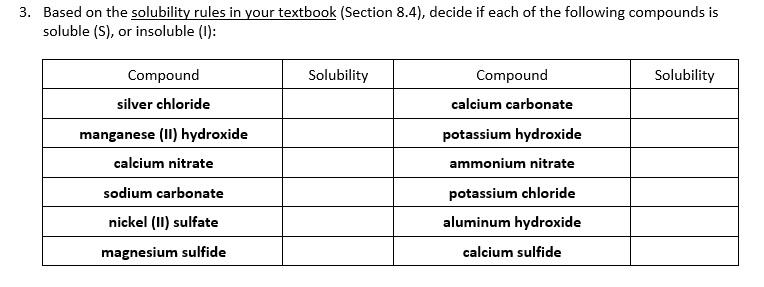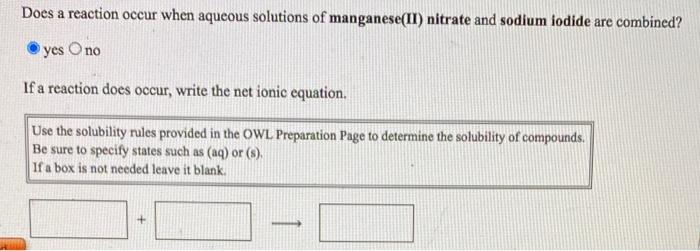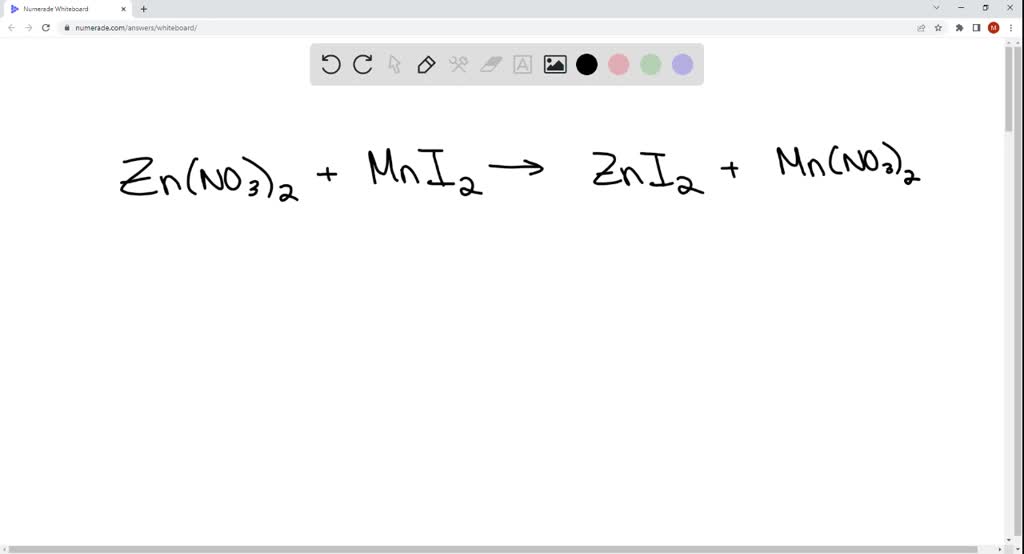
SOLVED: Does a reaction occur when aqueous solutions of zinc nitrate and manganese(II) iodide are combined? Yes, a reaction does occur. Write the net ionic equation. Use the solubility rules provided in

SOLVED: Use the solubility generalizations on the information page to predict if one or more precipitates will form when aqueous solutions of manganese(II) sulfate (MnSO4) and zinc nitrate (Zn(NO3)2) are mixed. Write
SOLVED: Does a reaction occur when aqueous solutions of manganese(II) nitrate and potassium hydroxide are combined? Yes/No. If a reaction does occur, write the net ionic equation. Use the solubility rules provided

Buy Manganese nitrate 99% Industrial Grade Colorless or rose red monoclinic crystal system Industrial Grade from Shandong Lingnuo chemical Co., Ltd - ECHEMI

TGA-DSC curves of Mn(NO 3 ) 2 $ 4H 2 O decomposition in air (heating... | Download Scientific Diagram