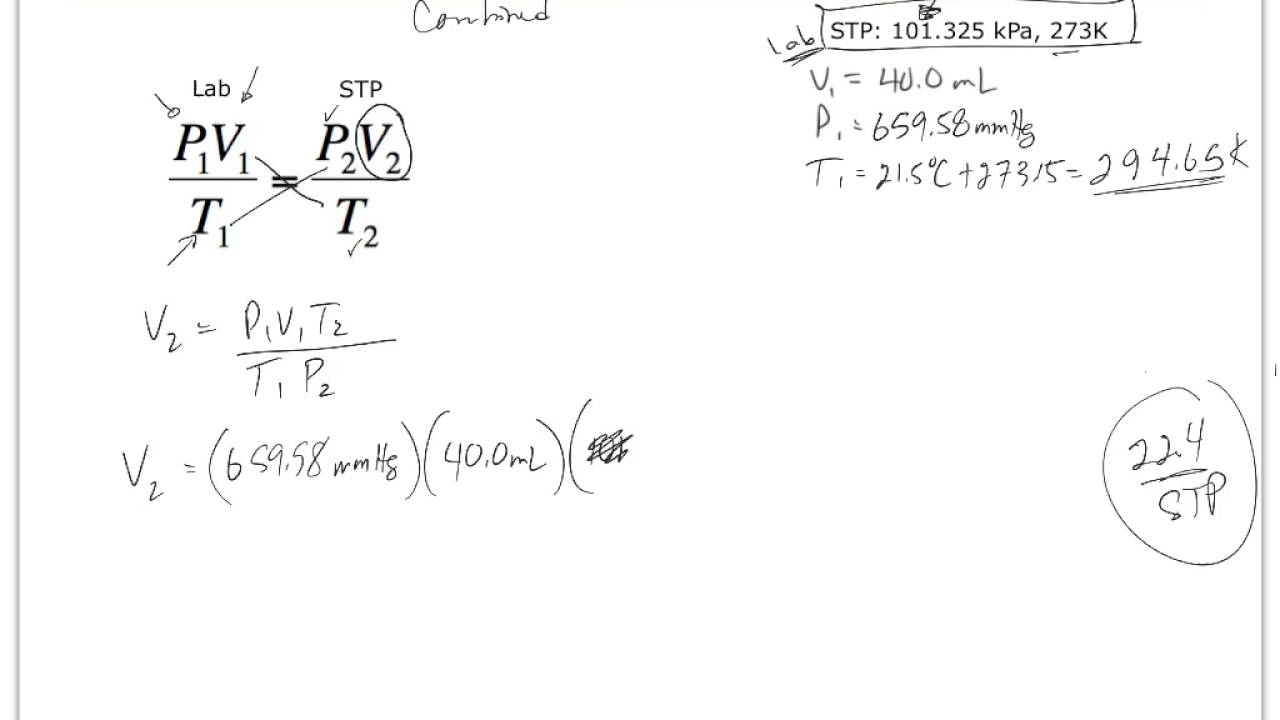SOLVED: Molar Volume of Hydrogen Data Table Trial 1 Trial 2 X 009439 X a.H8mL 8 .28-L 22.8 C Length of Mg Ribbon Mass of Mg 00942 g Evidence of Chemical Reaction

Sample Lab Report Molar volume of Hydrogen - Sample Lab Report: Molar Volume Of Hydrogen Background: - Studocu

SOLVED: In the experiment of determination of molar volume of Hydrogen gas, you are given the following data: Mass of Mg = 0.02 g. Volume of the gas in the calibrated part =

SOLVED: A student performed an experiment to determine the molar volume of hydrogen using the reaction between magnesium and excess hydrochloric acid. The following data was collected: - Room temperature: 21°C -

SOLVED: Molar Volume of Hydrogen Lab10 pdf alamo edu Lab10 pdf Molar Volume of Hydrogen Trial 1: 0.0199 mol Trial 2: 0.03 mol Length of Mg Ribbon: 3 cm Mass of Mg:
49. What will be the ratio of molar volume to atomic volume for 1mol. of hydrogen ? (Take the size of molecule to be 1Angstrom) ?

Molar volume of solid hydrogen as a function of pressure, together with... | Download Scientific Diagram