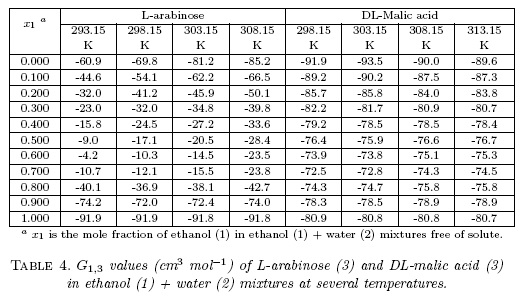
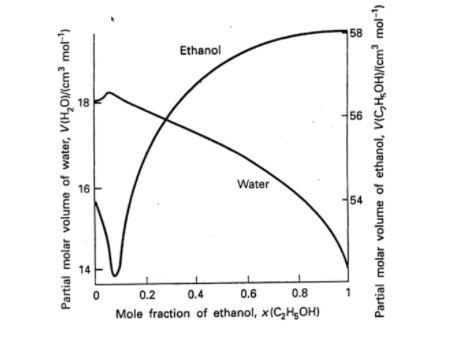
Excess molar volumes, VE, against the mole fraction of ethanol, x1, for... | Download Scientific Diagram

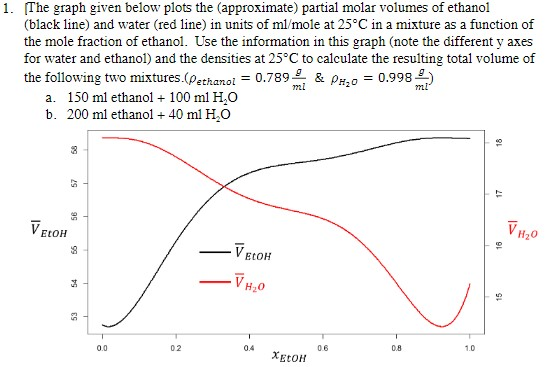
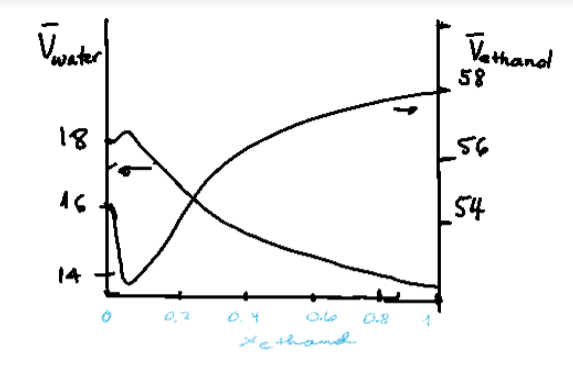
SOLVED: Using the graph below, the molar volume of ethanol and the partial molar volume of ethanol at 0.90 mole fraction of ethanol are: VEtoH = 58.4 mL/mol Vi,o = 56.1 mL/mol

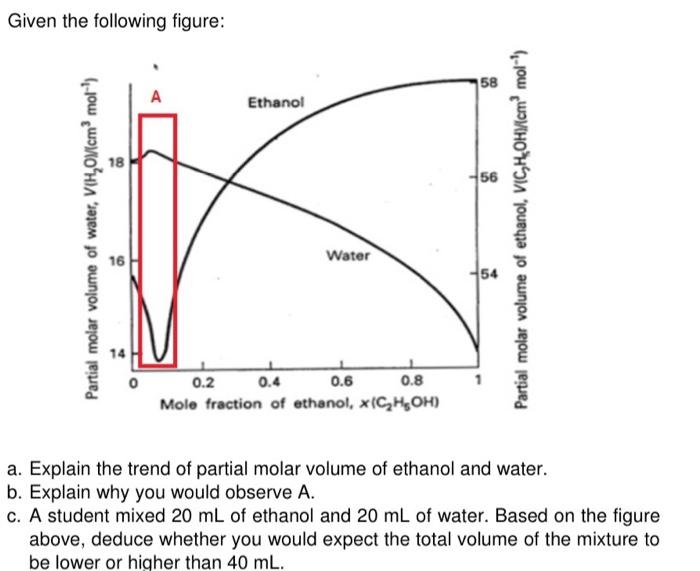
SOLVED: The partial molar volume for each component in a water/ethanol mixture is shown to the right: 58.12 cm³/mol Water If we mix 450 cm³ of ethanol with 550 cm³ of water,
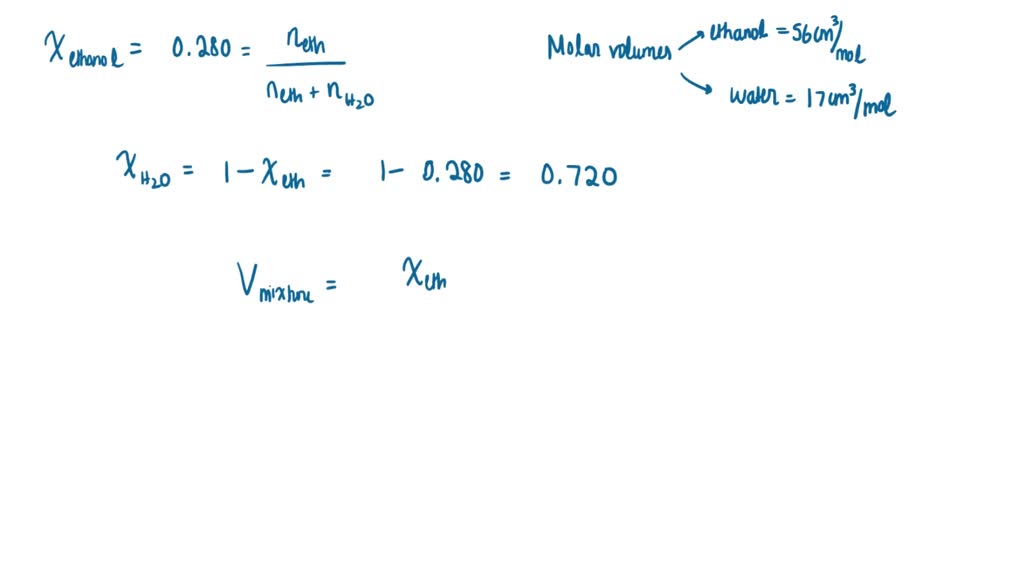
SOLVED: Calculate the volume of one mole (molar volume) of a mixture of water and ethanol in which mole fraction of ethanol is 0.280. Partial molar volumes of ethanol and water at

Excess molar volumes, V E , against the mole fraction of ethanol, x1,... | Download Scientific Diagram