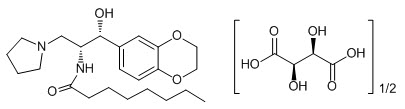
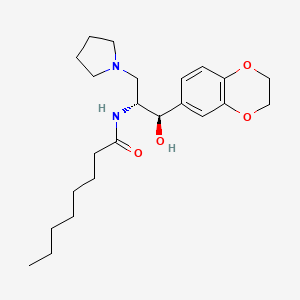
1 Louisiana Medicaid Enzyme Replacement Therapy The Louisiana Uniform Prescription Drug Prior Authorization Form should be uti

How a concentration-effect analysis of data from the eliglustat thorough electrocardiographic study was used to support dosing recommendations - ScienceDirect

Once- versus twice-daily dosing of eliglustat in adults with Gaucher disease type 1: The Phase 3, randomized, double-blind EDGE trial - ScienceDirect

Long-term adverse event profile from four completed trials of oral eliglustat in adults with Gaucher disease type 1 | Orphanet Journal of Rare Diseases | Full Text

A pooled analysis of adverse events in 393 adults with Gaucher disease type 1 from four clinical trials of oral eliglustat: Evaluation of frequency, timing, and duration - ScienceDirect
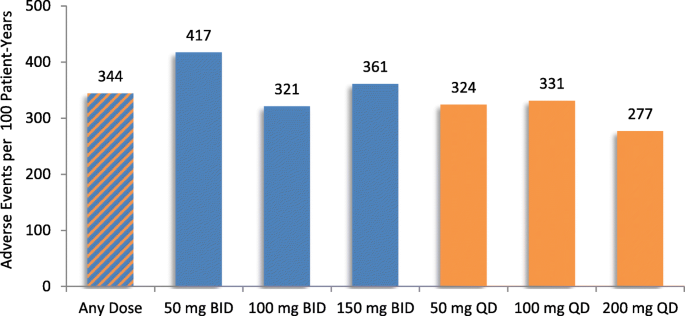
Long-term adverse event profile from four completed trials of oral eliglustat in adults with Gaucher disease type 1 | Orphanet Journal of Rare Diseases | Full Text