Why Australia is the Destination of Choice for International Medical Device Clinical Trials : MTPConnect

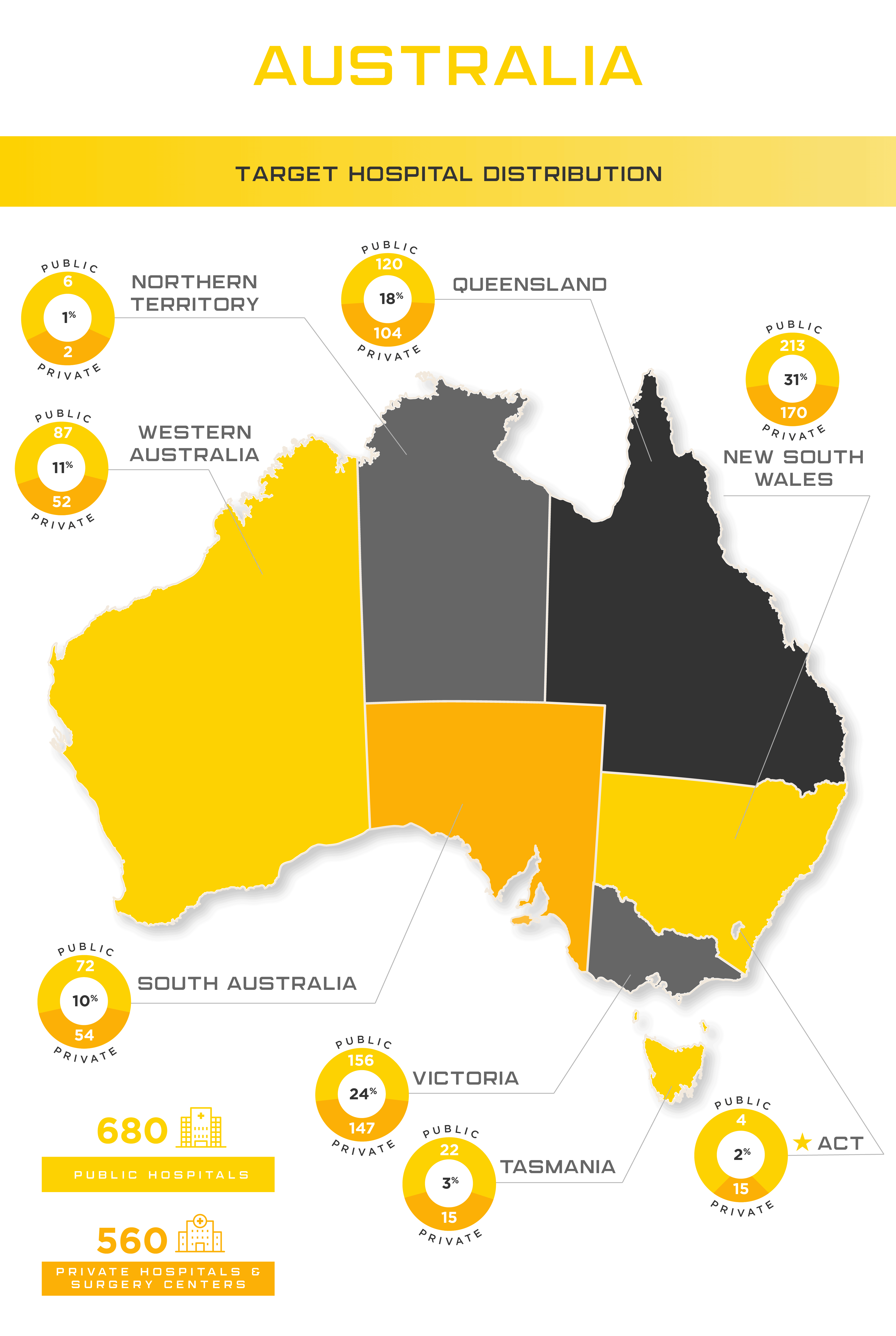
The regulation of medical devices in Australia. Overview Comparing medicines and medical devices What is a medical device? Statistics on patients requiring. - ppt download

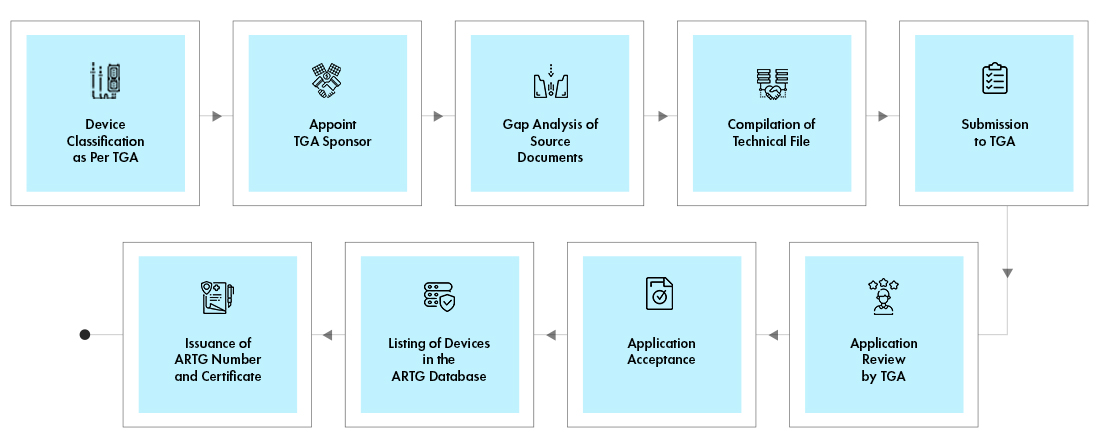
Newsletter January 2023 – Medical Device Regulations – update on Australian regulatory requirements for software based medical devices | Certification Body Australia (CBA)

The regulatory standards for the approval of medical devices in Australia | Australian Government Department of Health and Aged Care
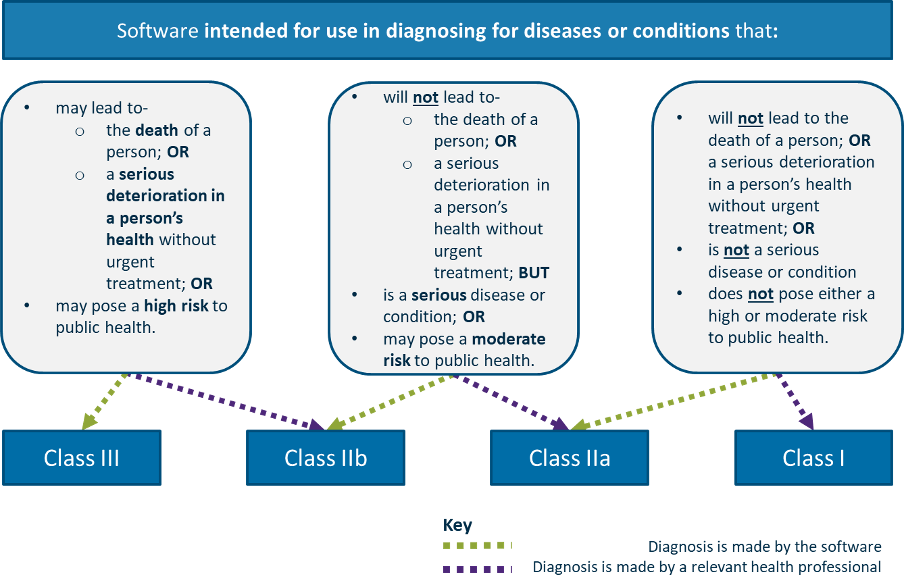
Hey, European Commission, it's time to copy-paste Australian regulation! - Software in Medical Devices, by MD101 Consulting