a syringe has a volume of 10.0 cm3 at pressure of 1 atmosphere if you plug the end so that no gas can escape - Brainly.in

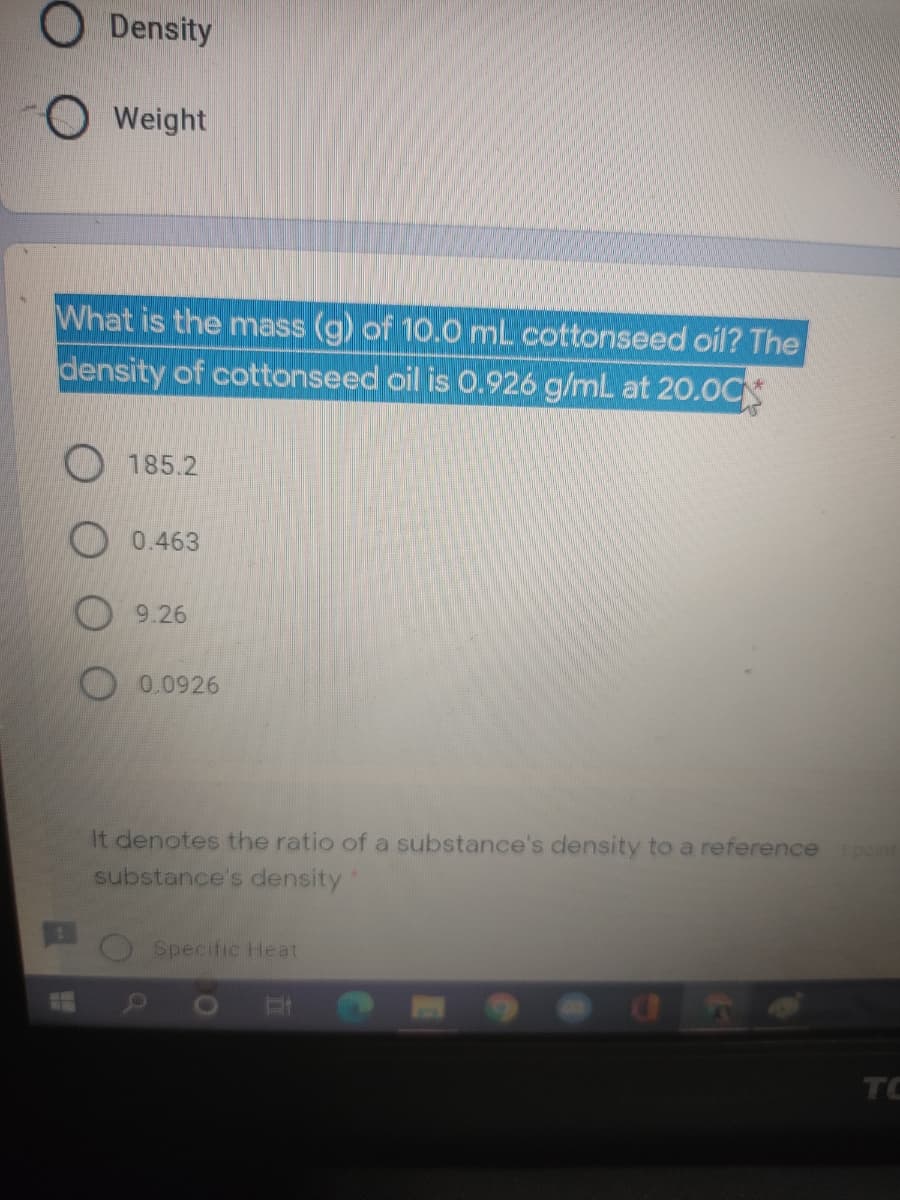
Solve the following. A syringe has a volume of 10.0 cm3 at pressure 1 atm. If you plug the end so that no gas can escape and push the plunger down, what

VIVEX: A Formula for Calculating Individual Vitreous Volume: A New Approach Towards Tailored Patient Dosing Regime in Intravitreal Therapy | Ophthalmology and Therapy

SOLVED: A syringe has a volume of 10.0 cm3 at pressure 1 atm. If you plug the end so that no gas can escape and push the plunger down, what must be

A syringe has a volume of 10.0cm^3 at pressure 1 atm.If you plug the end so that no gas can escape and push - Brainly.in
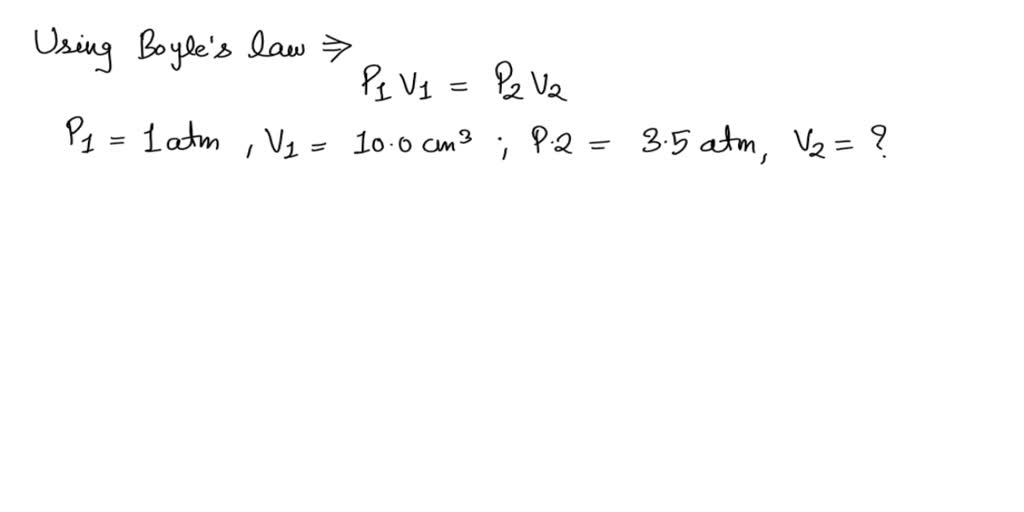
SOLVED: a syringe has a volume of 10.0 cm3 at pressure of 1 atmosphere if you plug the end so that no gas can escape and push the plunger down what must
A sample gas has a volume of 10 L and a pressure of 1 ATM What is the new pressure if the volume is decreased to 3L? - Quora
Solved: Example 2: A syringe has a volume of 10.0 ccs. The pressure is 1.0 atm. If you plug the en [Chemistry]

molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises
A syringe has a volume of 10.0 cm^3 at pressure 1 atm. If you plug the end so that no gas can escape and push the plunger down, - Sarthaks eConnect

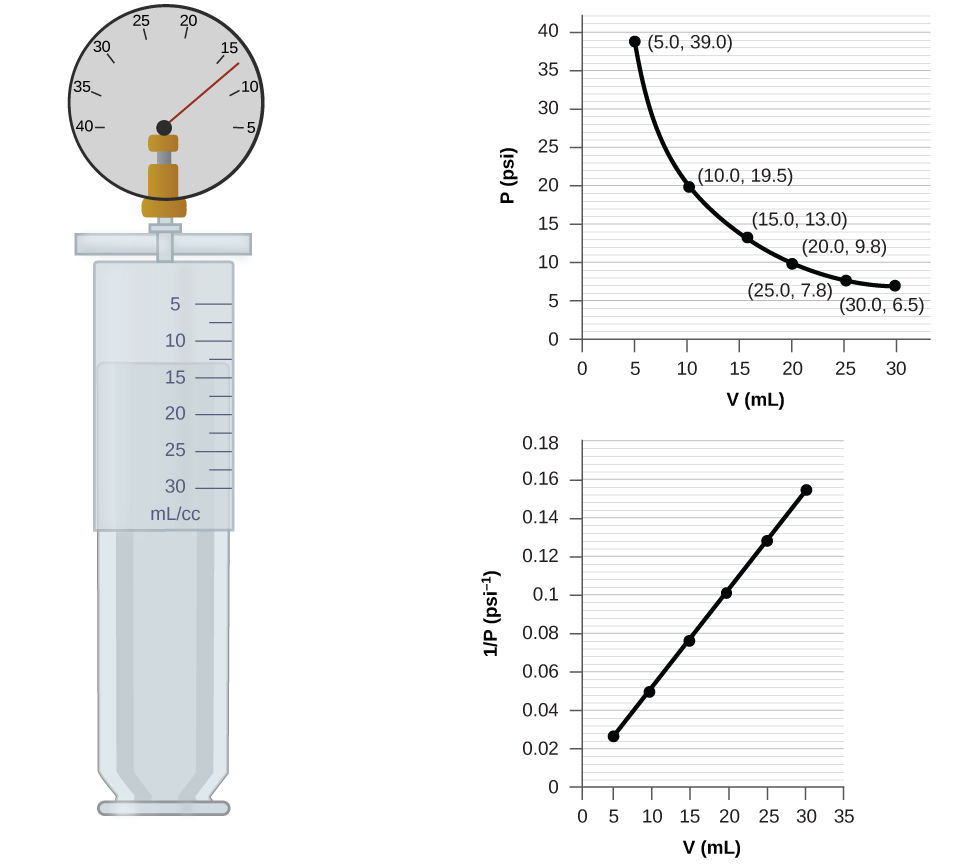
8.2: Relating Pressure, Volume, Amount, and Temperature: The Ideal Gas Law | General College Chemistry I





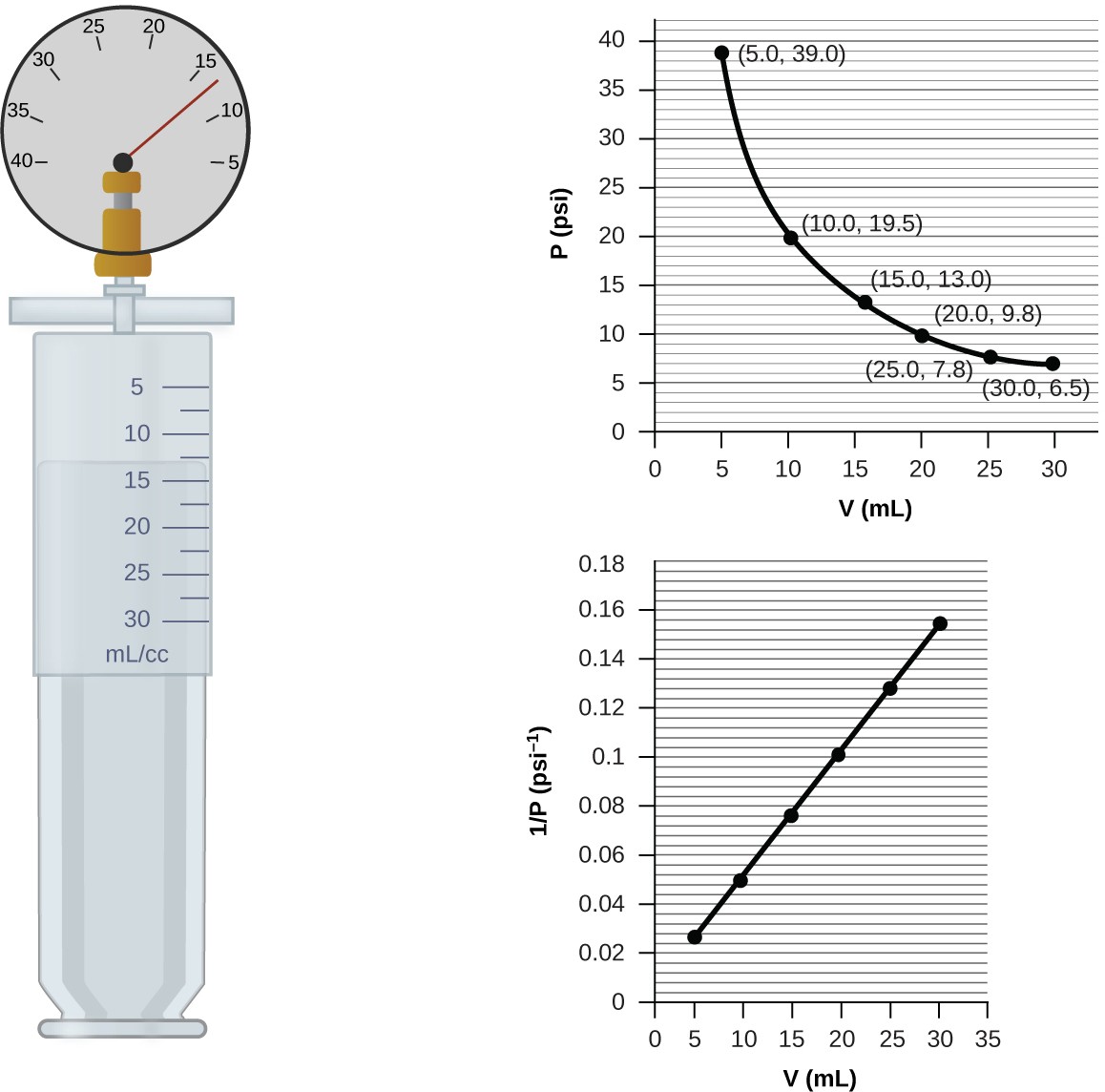

![Marathi] A syringe has a volume of 10.0 cm^3 at pressure 1 atm. If yo Marathi] A syringe has a volume of 10.0 cm^3 at pressure 1 atm. If yo](https://static.doubtnut.com/ss/web/11274255.webp)